Analysis of relationship between serum hypoxia inducible factor-1α, tenascin C and prognosis of hemodialysis patients with chronic renal failure based on decision curve
-
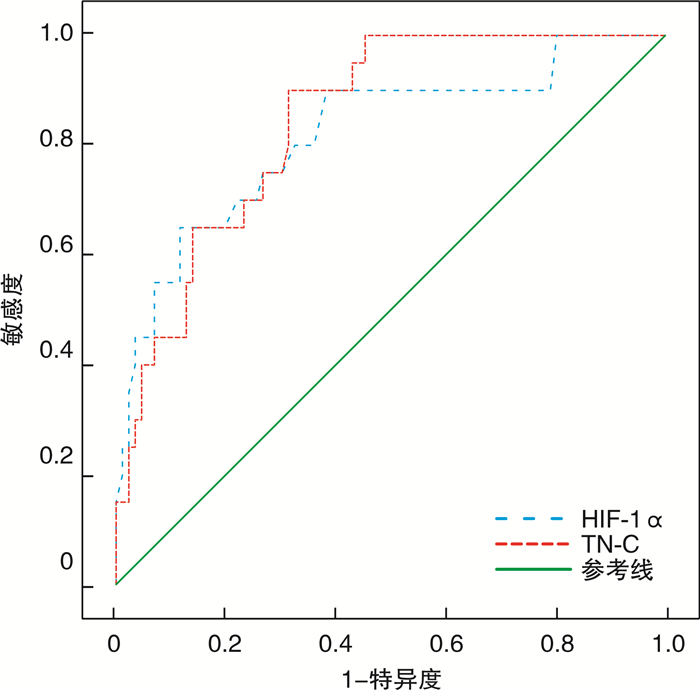
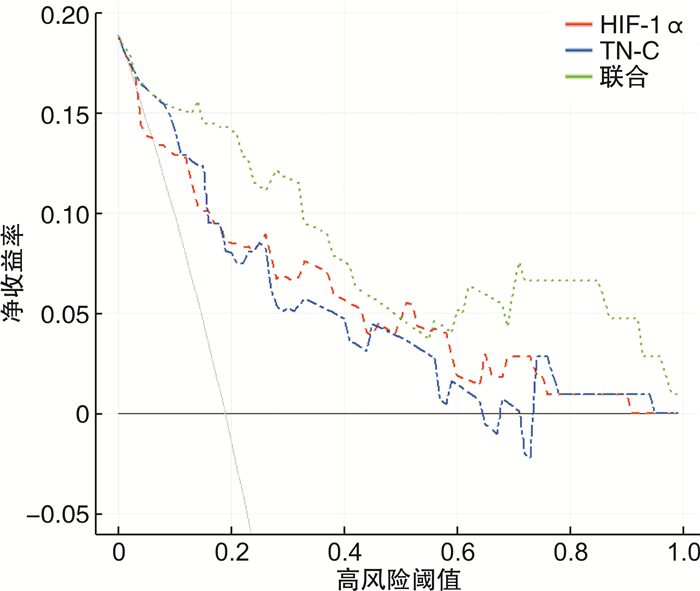
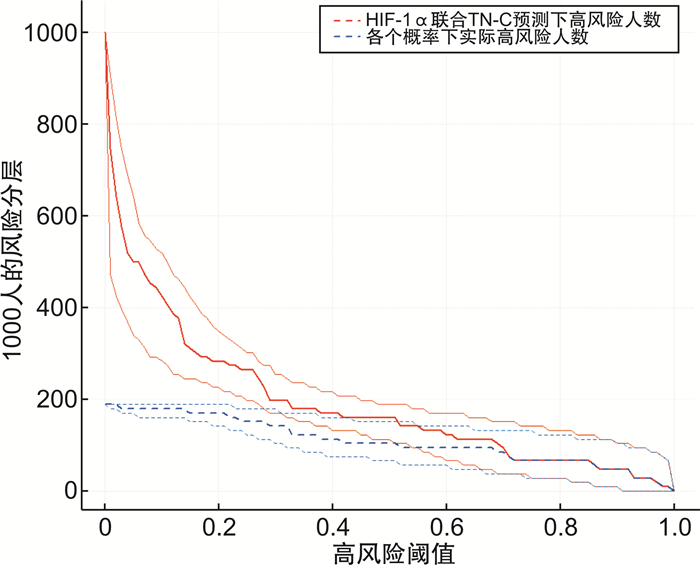
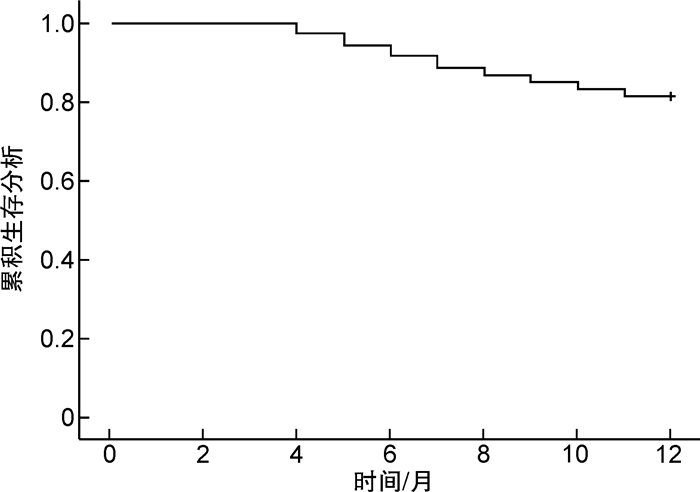
摘要: 目的 基于决策曲线分析血清缺氧诱导因子(HIF-1α)、腱糖蛋白C(TN-C)与慢性肾衰竭(CRF)进行血液透析(HD)患者预后的关系。方法 选择2020年7月至2021年7月106例CRF行HD治疗患者作为研究对象,测定患者入院时血清HIF-1α、TN-C水平,所有患者均接受每月1次随访,持续随访1年,记录患者预后情况,分为预后不良组(死亡)和预后良好组(存活),比较2组基线资料和入院时血清HIF-1α、TN-C水平,经Cox回归分析血清HIF-1α、TN-C水平对CRF患者行HD治疗预后的影响,绘制ROC曲线和决策曲线分析入院时血清HIF-1α、TN-C水平,预测CRF患者行HD治疗预后不良价值和净受益率。结果 随访1年统计106例进行HD治疗的CRF患者生存情况,以死亡为终点事件,随访时间1~12个月,20例死亡,预后不良率为18.87%(20/106)。经Kaplan-Meier法绘制生存曲线显示,20例死亡患者平均生存时间为(7.10±2.31)个月;预后不良组CRF患者病程长于预后良好组,透析充分性高于预后良好组,HIF-1α、TN-C水平高于预后良好组,差异有统计学意义(P< 0.05);经Cox回归分析显示,病程长、HIF-1α及TN-C水平高均可能是CRF患者行HD治疗预后不良的影响因素(P< 0.05);森林图发现HIF-1α与CRF患者HD治疗预后不良的关联较强;ROC曲线图结果显示,入院时血清HIF-1α、TN-C水平预测CRF患者HD治疗预后不良的AUC分别为0.819、0.845,均有一定预测价值;决策曲线结果显示,在阈值0~0.5,联合入院时血清HIF-1α、TN-C水平预测CRF患者HD治疗预后的净受益率优于单纯HIF-1α、TN-C,净受益率最大值为0.189。结论 血清HIF-1α、TN-C水平高与CRF患者HD治疗预后不良有关,联合入院时血清HIF-1α、TN-C水平预测CRF患者HD治疗预后价值较高,可获得较大净受益率。Abstract: Objective To analyze the relationship between serum hypoxia inducible factor-1α(HIF-1α), tenascin-C(TN-C) and the prognosis of hemodialysis(HD) patients with chronic renal failure(CRF) based on decision curve.Methods 106 CRF patients treated with HD in the hospital from July 2020 to July 2021 were selected as the research subjects, the levels of serum HIF-1α and TN-C of patients were measured at admission.All patients were followed up once a month, the follow-up was lasted for 1 year, the prognosis of patients was recorded and divided into poor prognosis group(death from illness) and good prognosis group(survival), the baseline data and serum HIF-1α and TN-C levels at admission were compared between the two groups, the effect of serum HIF-1α and TN-C levels on the prognosis of CRF patients with HD were analyzed by Cox regression, ROC curve and decision curve were drawn, analyzed the levels of serum HIF-1α and TN-C at admission, and predicted the value and net benefit rate of poor prognosis of HD in CRF patients.Results The survival of 106 CRF patients treated with HD was counted after 1 year of follow-up, the end event was death from illness, and the follow-up time was 1-12 months, 20 patients died from illness, the poor prognosis accounted for 18.87%(20/106). The survival curve drawn by Kaplan-Meier method showed that the average survival time of 20 patients dead from illness was(7.10±2.31) months; the course of CRF patients in poor prognosis group was longer than that in good prognosis group, the adequacy of dialysis was higher than that in good prognosis group, and the levels of HIF-1α and TN-C were higher than those in good prognosis group, with statistical significant differences(P< 0.05); the results of Cox regression analysis showed that, long course of disease and high levels of HIF-1α and TN-C might be the influencing factors of poor prognosis in patients with CRF after HD treatment(P< 0.05); forest map was showed that HIF-1α was strongly associated with poor prognosis of HD treatment in CRF patients; the ROC curve showed that the AUC of serum HIF-1α and TN-C at admission in predicting the poor prognosis of CRF patients with HD were 0.819 and 0.845, respectively, which had certain predictive value; the decision curve showed that within the threshold range of 0-0.5, the net benefit rate of combining serum HIF-1α and TN-C levels at admission in predicting the prognosis of HD in CRF patients was better than that of HIF-1α and TN-C alone, the maximum net benefit rate was 0.189.Conclusion The high levels of serum HIF-1α and TN-C are related to the poor prognosis of HD treatment in CRF patients, combined with the levels of serum HIF-1α and TN-C at admission, it had a high value in predicting the prognosis of HD treatment in CRF patients, and can obtain a large net benefit rate.
-
Key words:
- chronic renal failure /
- hemodialysis /
- hypoxia inducible factor-1α /
- tenascin C
-

-
表 1 不同预后的HD治疗CRF患者基线资料、实验室指标比较
X ±S 因素 预后不良组(20例) 预后良好组(86例) χ2/t P 性别/例(%) 0.220 0.639 男 13(65.00) 51(59.30) 女 7(35.00) 35(40.70) 年龄/岁 50.60±5.80 51.65±5.54 0.758 0.450 病程/年 2.80±0.52 2.43±0.54 2.760 0.007 原发病/例(%) 0.570 0.913 肾小球肾炎 12(60.00) 50(58.14) 糖尿病肾病 5(25.00) 18(20.93) 高血压肾病 2(10.00) 10(11.63) 其他 1(5.00) 8(9.30) 透析充分性/例(%) 9.582 0.002 不充分 12(60.00) 21(24.42) 充分 8(40.00) 65(75.58) 血红蛋白/(g·L-1) 103.82±10.38 101.51±11.34 0.831 0.408 血清白蛋白/(g·L-1) 22.02±2.27 23.16±2.66 1.778 0.078 空腹血糖/(mmol·L-1) 7.29±0.76 7.08±0.72 1.157 0.250 血肌酐/(μmol·L-1) 742.26±76.55 736.24±77.27 0.314 0.754 尿素氮/(mmol·L-1) 31.19±3.09 31.81±3.14 0.794 0.429 HIF-1α/(μg·L-1) 1.69±0.31 1.31±0.28 5.353 < 0.001 TN-C/(μg·L-1) 65.58±6.32 56.14±6.78 5.678 < 0.001 表 2 CRF患者HD治疗预后情况影响因素Cox回归分析
因素 B SE Wald P HR 95%CI 病程 1.033 0.520 3.942 0.047 2.809 1.013~7.788 透析充分性 0.895 0.486 3.391 0.066 2.447 0.944~6.345 HIF-1α 1.776 0.725 6.001 0.014 5.903 1.426~24.437 TN-C 0.103 0.034 9.502 0.002 1.109 1.038~1.184 表 3 入院时血清HIF-1α、TN-C水平预测CRF患者HD治疗预后
因素 AUC cut-off 95%CI P 特异度 灵敏度 约登指数 HIF-1α 0.819 1.485 0.707~0.931 < 0.001 0.733 0.750 0.483 TN-C 0.845 59.625 0.765~0.925 < 0.001 0.686 0.900 0.586 -
[1] 陈静, 陈奕鸣, 杨晶, 等. 维持性血液透析患者血浆正五聚蛋白的变化与心血管事件危险因素的相关性[J]. 中国血液净化, 2020, 19(3): 183-186. https://www.cnki.com.cn/Article/CJFDTOTAL-ZGJH202003011.htm
[2] Wong B, Ravani P, Oliver MJ, et al. Comparison of Patient Survival Between Hemodialysis and Peritoneal Dialysis Among Patients Eligible for Both Modalities[J]. Am J Kidney Dis, 2018, 71(3): 344-351. doi: 10.1053/j.ajkd.2017.08.028
[3] Zhang J, Zhang Q, Lou Y, et al. Hypoxia-inducible factor-1α/interleukin-1β signaling enhances hepatoma epithelial-mesenchymal transition through macrophages in a hypoxic-inflammatory microenvironment[J]. Hepatology, 2018, 67(5): 1872-1889. doi: 10.1002/hep.29681
[4] 冉茂霞, 康婷, 朱婷婷, 等. 缺氧诱导因子1α信号通路在慢性肾脏病血管钙化大鼠主动脉中的表达及作用[J]. 中华肾脏病杂志, 2021, 37(9): 749-757. doi: 10.3760/cma.j.cn441217-20200831-00110
[5] Zhu H, Liao J, Zhou X, et al. Tenascin-C promotes acute kidney injury to chronic kidney disease progression by impairing tubular integrity via αvβ6 integrin signaling[J]. Kidney Int, 2020, 97(5): 1017-1031. doi: 10.1016/j.kint.2020.01.026
[6] 张新研. 外周血及尿液中相关指标在中老年早期糖尿病肾病中的研究[J]. 临床血液学杂志, 2021, 34(8): 564-567. https://www.cnki.com.cn/Article/CJFDTOTAL-LCXZ202108009.htm
[7] 中华中医药学会. 慢性肾衰竭诊疗指南[J]. 中国中医药现代远程教育, 2011, 9(9): 132-133. https://www.cnki.com.cn/Article/CJFDTOTAL-ZZXJ201509001.htm
[8] 刘文虎, 孙雪峰, 林洪丽, 等. 中国血液透析充分性临床实践指南[J]. 中华医学杂志, 2015, 95(34): 2748-2753. doi: 10.3760/cma.j.issn.0376-2491.2015.34.004
[9] Cobo G, Lindholm B, Stenvinkel P. Chronic inflammation in end-stage renal disease and dialysis[J]. Nephrol Dial Transplant, 2018, 33: iii35-iii40. doi: 10.1093/ndt/gfy175
[10] Klinger M, Madziarska K. Mortality predictor pattern in hemodialysis and peritoneal dialysis in diabetic patients[J]. Adv Clin Exp Med, 2019, 28(1): 133-135.
[11] 寇宏博, 韩冬冰, 范以生, 等. 缺氧诱导因子1α、上皮钙粘附蛋白在肾透明细胞癌中表达及临床意义[J]. 临床军医杂志, 2020, 48(6): 649-652. https://www.cnki.com.cn/Article/CJFDTOTAL-JYGZ202006010.htm
[12] He Y, Lang X, Cheng D, et al. Curcumin Ameliorates Chronic Renal Failure in 5/6 Nephrectomized Rats by Regulation of the mTOR/HIF-1α/VEGF Signaling Pathway[J]. Biol Pharm Bull, 2019, 42(6): 886-891. doi: 10.1248/bpb.b18-00787
[13] 杨军, 万晨阳, 李婷, 等. 血清可溶性生长刺激基因表达蛋白2、腱糖蛋白C水平与维持性血液透析患者并发心血管事件的关系[J]. 中国血液净化, 2020, 19(6): 376-380. doi: 10.3969/j.issn.1671-4091.2020.06.005
[14] 刘媛, 隋小妮, 王安阳, 等. 腹膜透析患者血清肌腱蛋白-C与腹主动脉钙化的相关研究[J]. 中国全科医学, 2020, 23(3): 312-318. https://www.cnki.com.cn/Article/CJFDTOTAL-QKYX202003013.htm
[15] 陈豫闽, 刘俊英, 贾国强, 等. 血清转化生长因子β_1、单核细胞趋化蛋白-1、低氧诱导因子-1α水平与慢性肾衰竭患者肠道菌群的相关性[J]. 临床内科杂志, 2021, 38(9): 605-608. doi: 10.3969/j.issn.1001-9057.2021.09.009
[16] Wang Y, Zhang L, Jin H, et al. Based on HIF-1α/Wnt/β-Catenin Pathway to Explore the Effect of Qingshen Granules on Chronic Renal Failure Patients: A Randomized Controlled Trial[J]. Evid Based Complement Alternat Med, 2019, 2019: 7656105.
[17] 赵波, 陈曦, 李洪波, 等. HIF1α调控TRPC6通道在肾小球系膜细胞缺氧损伤中的分子机制[J]. 中国中西医结合肾病杂志, 2018, 19(8): 668-671. doi: 10.3969/j.issn.1009-587X.2018.08.004
[18] 李富强. 血液透析和腹膜透析对慢性肾衰竭尿毒症患者微炎症状态的影响及与心血管疾病的关系[J]. 临床内科杂志, 2019, 36(11): 748-750. doi: 10.3969/j.issn.1001-9057.2019.11.010
[19] 冯敬芳, 王雯瑾, 饶龙华. 维持性血液透析患者血清HIF-1α VEGF水平与冠状动脉钙化的相关性[J]. 河北医学, 2019, 25(5): 759-763. doi: 10.3969/j.issn.1006-6233.2019.05.12
[20] Chen S, Fu H, Wu S, et al. Tenascin-C protects against acute kidney injury by recruiting Wnt ligands[J]. Kidney Int, 2019, 95(1): 62-74. doi: 10.1016/j.kint.2018.08.029
[21] 高振宇, 马艳, 祝文静. 血清sST2、TN-C水平与维持性血液透析患者心血管事件发生的关系[J]. 山东医药, 2020, 60(31): 49-52. https://www.cnki.com.cn/Article/CJFDTOTAL-SDYY202031012.htm
[22] Broeker K, Fuchs M, Schrankl J, et al. Different subpopulations of kidney interstitial cells produce erythropoietin and factors supporting tissue oxygenation in response to hypoxia in vivo[J]. Kidney Int, 2020, 98(4): 918-931.
[23] Fu H, Tian Y, Zhou L, et al. Tenascin-C Is a Major Component of the Fibrogenic Niche in Kidney Fibrosis[J]. J Am Soc Nephrol, 2017, 28(3): 785-801.
[24] Ebrahimi V, Khademian MH, Masoumi SJ, et al. Factors influencing survival time of hemodialysis patients; time to event analysis using parametric models: a cohort study[J]. BMC Nephrol, 2019, 20(1): 215.
-




 下载:
下载: