Effects of regulatory T cell and human leukocyte antigen class Ⅱ molecule expression in anaemia patients on haemoglobin
-
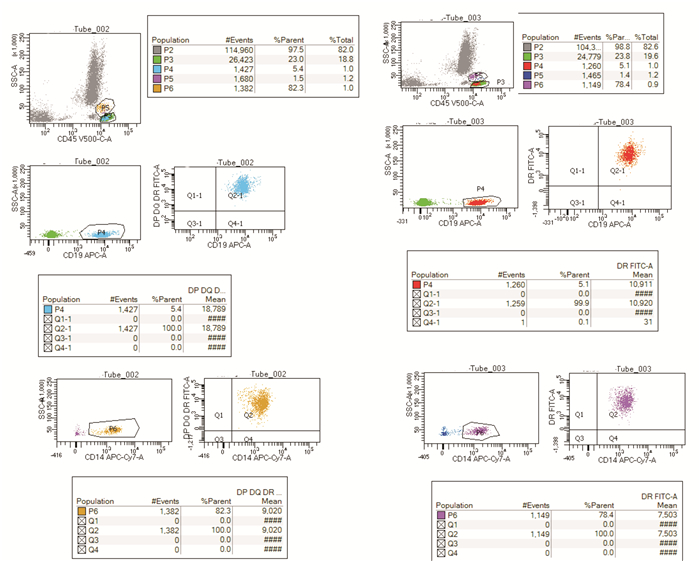
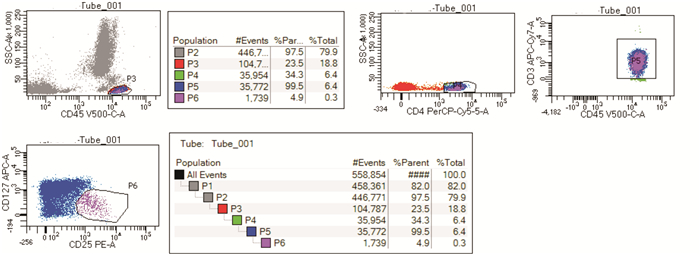
摘要: 目的人类白细胞抗原Ⅱ(HLA-Ⅱ)由抗原提呈细胞表达直接参与T细胞活化,调节性T细胞(Treg)在免疫系统中发挥调节免疫反应的重要作用。贫血是血液病学常见的疾病,病因众多。本文探讨不同贫血患者与HLA-Ⅱ分子表达及Treg细胞比例之间的相关性。方法以自身免疫性溶血性贫血(AIHA)、骨髓衰竭性疾病(BMFS)、B细胞-浆细胞肿瘤侵犯骨髓、继发于感染或肝肾疾病的慢性病贫血患者为研究对象,检测以上疾病患者HLA-Ⅱ分子表达强度、Treg细胞比例及与贫血程度的相关性。结果所有贫血患者的HLA-Ⅱ类分子表达在淋巴细胞和单核细胞中差异无统计学意义;AIHA患者血红蛋白含量与外周血Treg细胞比例呈负相关;BMFS患者Treg细胞比例与淋巴细胞HLA-Ⅱ表达呈正相关;B细胞-浆细胞肿瘤侵犯骨髓患者血红蛋白含量与淋巴细胞HLA-Ⅱ及HLA-DR表达呈正相关;继发于感染或肝肾疾病的慢性病贫血患者血红蛋白含量与单核细胞HLA-Ⅱ及HLA-DR表达呈负相关,外周血Treg细胞比例与淋巴细胞HLA-Ⅱ及HLA-DR表达呈正相关。结论AIHA、BMFS、B细胞-浆细胞肿瘤侵犯骨髓、继发于感染或肝肾疾病的慢性病贫血患者的单核细胞免疫反应较健康供者活跃;B细胞-浆细胞肿瘤累及骨髓患者的恶性淋巴细胞通过低表达HLA分子来实现免疫逃逸及大量增殖;继发于感染或肝肾疾病的慢性病贫血促进单核细胞的活化,其Treg细胞比例与淋巴细胞HLA-Ⅱ分子表达相关;Treg细胞减少参与AIHA的发生及发展,与外周血主要抗原提呈细胞的HLA-Ⅱ类分子及HLA-DR分子表达无明显相关;BMFS患者Treg细胞比例与淋巴细胞HLA-Ⅱ分子表达呈正相关,而与淋巴细胞HLA-DR分子表达不相关,提示其发病机制尤其复杂。Abstract: ObjectiveHLA-Ⅱ molecules are directly involved in T-cell activation through antigen presenting cell expression. Regulatory T cells(Tregs) play an important role in regulating systemic immune responses. Anaemia is a common haematological disease with many causes. This study is to identify a possible correlation between HLA-Ⅱ molecular expression and Treg cell ratio.MethodsIn this study, patients with autoimmune haemolytic anaemia(AIHA), bone marrow failure disease(BMFS), B-plasma cell tumour invading bone marrow, chronic anaemia secondary to infection, or liver and kidney diseases were studied to detect HLA-Ⅱ molecular expression and Treg cell ratio, and identify a possible correlation with anaemia severity.ResultsThere was no significant difference in the expression of HLA-Ⅱ molecules in lymphocytes and monocytes in all patients with anemia; the hemoglobin content in AIHA patients was negatively correlated with the proportion of Treg in peripheral blood; the proportion of Treg in BMFS patients was positively correlated with the expression of HLA-Ⅱ in lymphocytes; the hemoglobin content of patients with B-plasma cell tumours invading the bone marrow was positively correlated with the expression of HLA-Ⅱ and HLA-DR in lymphocytes; in patients with anemia of chronic disease secondary to infection or liver and kidney disease, the content of hemoglobin was negatively correlated with the expression of HLA-Ⅱ and HLA-DR in monocytes, the proportion of Treg in peripheral blood was positively correlated with the expression of HLA-Ⅱ and HLA-DR in lymphocytes.ConclusionThe monocyte immune response of patients with AIHA, BMFS, B-plasma cell tumour invading the bone marrow, and chronic anaemia secondary to infection or liver and kidney diseases was more active than that of healthy donors. The malignant lymphocytes of patients with B-plasma cell tumours involving bone marrow might realise immune escape and proliferation through low expression of HLA molecules. Anaemia secondary to infection or liver and kidney diseases might promote the activation of monocytes, and the Treg ratio might be related to the expression of HLA-Ⅱ molecules in lymphocytes. Treg reduction might be involved in the development and progression of AIHA and have no obvious correlation with the expression of HLA-Ⅱ and HLA-DR molecules in the main antigen presenting cells. The proportion of Tregs in patients with BMFS positively correlated with HLA-Ⅱ expression in lymphocytes, but not with HLA-DR expression in lymphocytes, suggesting that its pathogenesis might be particularly complicated.
-
Key words:
- anaemia /
- HLA-Ⅱ expression /
- HLA-DR expression /
- regulatory T cells
-

-
[1] Nada AM, Hammouda M. Immunoregulatory T cells, LFA-3 and HLA-DR in autoimmune thyroid diseases[J]. Indian J Endocrinol Metab, 2014, 18(4): 574-581. doi: 10.4103/2230-8210.137524
[2] Jevremovic D, Nanaa A, Geyer SM, et al. Abnormal CD13/HLA-DR Expression Pattern on Myeloblasts Predicts Development of Myeloid Neoplasia in Patients With Clonal Cytopenia of Undetermined Significance[J]. Am J Clin Pathol, 2022, 158(4): 530-536. doi: 10.1093/ajcp/aqac083
[3] Tsuji N, Hosokawa K, Urushihara R, et al. Frequent HLA-DR loss on hematopoietic stem progenitor cells in patients with cyclosporine-dependent aplastic anemia carrying HLA-DR15[J]. Leukemia, 2022, 36(6): 1666-1675. doi: 10.1038/s41375-022-01549-6
[4] Gao YL, Yao Y, Zhang X, et al. Regulatory T Cells: Angels or Demons in the Pathophysiology of Sepsis?[J]. Front Immunol, 2022, 13: 829210. doi: 10.3389/fimmu.2022.829210
[5] Chevalier N, Mueller M, Mougiakakos D, et al. Analysis of dendritic cell subpopulations in follicular lymphoma with respect to the tumor immune microenvironment[J]. Leuk Lymphoma, 2016, 57(9): 2150-2160. doi: 10.3109/10428194.2015.1135432
[6] Saunthararajah Y, Nakamura R, Nam JM, et al. HLA-DR15(DR2) is overrepresented in myelodysplastic syndrome and aplastic anemia and predicts a response to immunosuppression in myelodysplastic syndrome[J]. Blood, 2002, 100(5): 1570-1574. doi: 10.1182/blood.V100.5.1570.h81702001570_1570_1574
[7] Boegel S, Löwer M, Bukur T, et al. HLA and proteasome expression body map[J]. BMC Med Genomics, 2018, 11(1): 36. doi: 10.1186/s12920-018-0354-x
[8] Jesionek-Kupnicka D, Bojo M, Prochorec-Sobieszek M, et al. HLA-G and MHC Class Ⅱ Protein Expression in Diffuse Large B-Cell Lymphoma[J]. Arch Immunol Ther Exp(Warsz), 2016, 64(3): 225-240. doi: 10.1007/s00005-015-0372-8
[9] de Las Cuevas Allende R, Díaz de Entresotos L, Conde Díez S. Anaemia of chronic diseases: Pathophysiology, diagnosis and treatment[J]. Med Clin(Barc), 2021, 156(5): 235-242.
[10] Petz LD, Garratty G. Immune Hemolytic Anemias[M]. 2nd ed. Philadelphia, Churchill Livingstone, 2004: 1-448.
[11] 曲莹莹, 邢莉民, 邵宗鸿. T淋巴细胞在自身免疫性溶血性贫血发病机制中的作用[J]. 国际输血及血液学杂志, 2017, 40(5): 450-454. doi: 10.3760/cma.j.issn.1673-419X.2017.05.016
[12] Ward FJ, Hall AM, Cairns LS, et al. Clonal regulatory T cells specific for a red blood cell autoantigen in human autoimmune hemolytic anemia[J]. Blood, 2008, 111(2): 680-687. doi: 10.1182/blood-2007-07-101345
[13] Ahmad E, Elgohary T, Ibrahim H. Naturally occurring regulatory T cells and interleukins 10 and 12 in the pathogenesis of idiopathic warm autoimmune hemolytic anemia[J]. J Investig Allergol Clin Immunol, 2011, 21(4): 297-304.
[14] Barker RN, Elson CJ. Multiple self epitopes on the rhesus polypeptide stimulate immunologically ignorant human T cells[J]. Eur J Immunol. 1994, 24: 1578-1582. doi: 10.1002/eji.1830240719
[15] Elson CJ, Barker RN, Thompson SJ, et al. Immunologically ignorant autoreactive T cells, epitope spreading and repertoire limitation[J]. Immunol Today, 1995, 16(2): 71-76. doi: 10.1016/0167-5699(95)80091-3
[16] Jersild C, Fog T, Hansen GS, et al. Histocompatibility determinants in multiple sclerosis, with special reference to clinical course[J]. Lancet, 1973, 2(7840): 1221-1225.
[17] Sprent J, Surh CD. Normal T cell homeostasis: the conversion of naive cells into memory-phenotype cells[J]. Nat Immunol, 2011, 12(6): 478-484. doi: 10.1038/ni.2018
[18] Markovic-Plese S, Cortese I, Wandinger KP, et al. CD4+CD28-costimulation-independent T cells in multiple sclerosis[J]. J Clin Invest, 2001, 108(8): 1185-1194. doi: 10.1172/JCI200112516
[19] Bielekova B, Sung MH, Kadom N, et al. Expansion and functional relevance of high-avidity myelin-specific CD4+ T cells in multiple sclerosis[J]. J Immunol, 2004, 172(6): 3893-3904. doi: 10.4049/jimmunol.172.6.3893
[20] Viglietta V, Baecher-Allan C, Weiner HL, et al. Loss of functional suppression by CD4+CD25+regulatory T cells in patients with multiple sclerosis[J]. J Exp Med, 2004, 199(7): 971-979. doi: 10.1084/jem.20031579
[21] Laplaud DA, Ruiz C, Wiertlewski S, et al. Blood T-cell receptor beta chain transcriptome in multiple sclerosis. Characterization of the Tcells with altered CDR3 length distribution[J]. Brain, 2004, 127(5): 981-995. doi: 10.1093/brain/awh119
[22] Mohme M, Hotz C, Stevanovic S, et al. HLA-DR15-derived self-peptides are involved in increased autologous T cell proliferation in multiple sclerosis[J]. Brain, 2013, 136(Pt 6): 1783-1798.
[23] Ooi JD, Petersen J, Tan YH, et al. Dominant protection from HLA-linked autoimmunity by antigen-specific regulatory T cells[J]. Nature, 2017, 545(7653): 243-247. doi: 10.1038/nature22329
[24] Phelps RG, Rees AJ. The HLA complex in Goodpasture's disease: a model for analyzing susceptibility to autoimmunity[J]. Kidney Int, 1999, 56(5): 1638-1653. doi: 10.1046/j.1523-1755.1999.00720.x
[25] 曹蓝, 洪鸣. 骨髓增生异常综合征/骨髓增殖性肿瘤的治疗进展[J]. 临床血液学杂志, 2021, 34(9): 680-683. doi: 10.13201/j.issn.1004-2806.2021.09.017 http://lcxz.cbpt.cnki.net/WKC/WebPublication/paperDigest.aspx?paperID=2ec11d98-e4c0-4215-b78b-4444f99e2bdb
[26] Lee W, Lee GR. Transcriptional regulation and development of regulatory T cells[J]. Exp Mol Med, 2018, 50(3): e456. doi: 10.1038/emm.2017.313
-




 下载:
下载: