Clinical characteristics and prognostic factors of 48 cases of primary systemic anaplastic large cell lymphoma
-
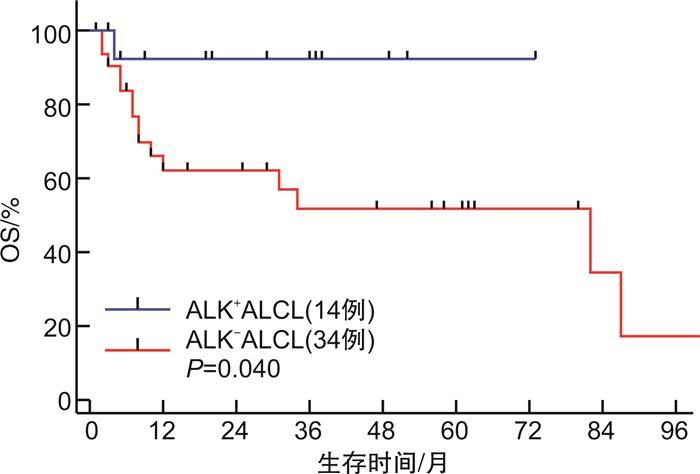
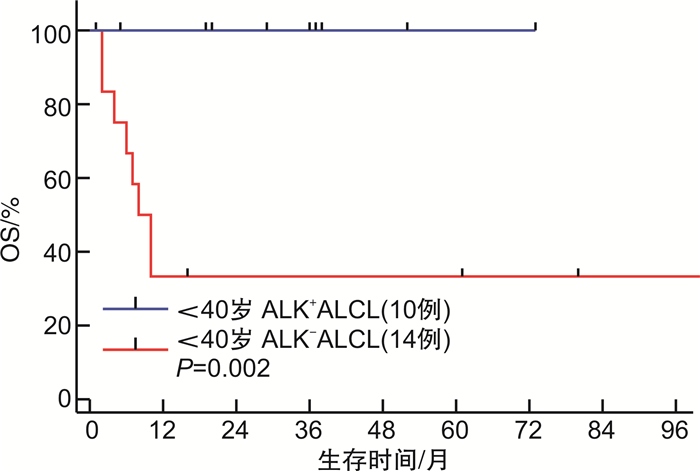
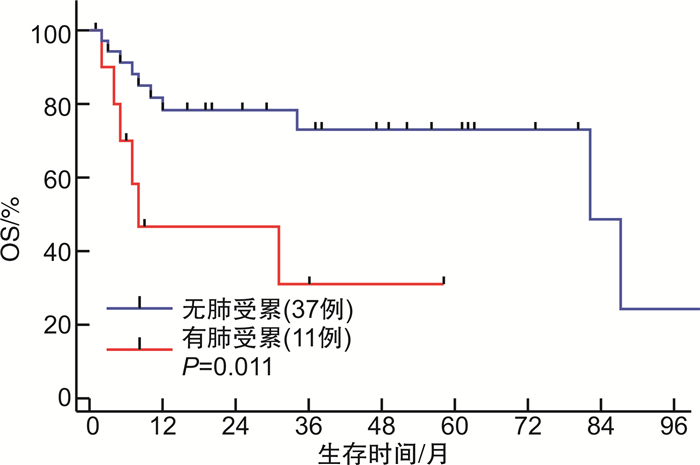
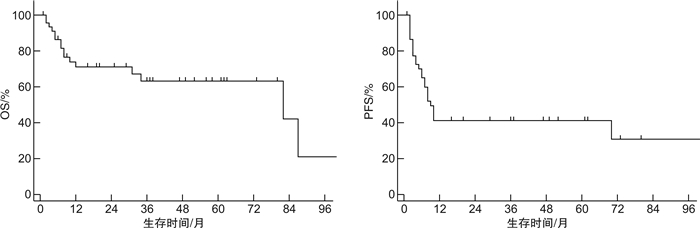
摘要: 目的 探讨原发系统性间变大细胞淋巴瘤(ALCL)患者的临床特征及预后影响因素。方法 回顾性分析2013年1月—2021年3月于北京协和医院诊治的48例ALCL患者的临床资料。结果 48例患者中男31例,女17例,中位发病年龄37(18~79)岁。ALK+ALCL 14例(29.2%),ALK-ALCL 34例(70.8%),Ann Arbor分期Ⅲ~Ⅳ期患者比例达79.2%,68.8%存在B症状,结外器官受累常见,54.2%存在>1个结外器官受累。48例患者中46例接受化疗,一线化疗方案包括GDP-ML方案6例,CHOEP/CHOP方案27例,西达本胺联合CHOP/CHOEP方案10例,维布妥昔单抗联合CHP方案3例,一线化疗完全缓解(CR)率为57.8%,总有效率为71.1%。中位随访时间为37(1~101)个月,整组患者1年及3年无进展生存率均为41.2%,1年及3年总生存(OS)率分别为71.1%和63.2%。ALK+ALCL组相比ALK-ALCL组除发病年龄更为年轻外(中位发病年龄25岁vs 50岁),其余临床特征相似,ALK+ALCL组一线化疗的CR率及OS率均显著优于ALK-ALCL组(CR率:85.7% vs 41.2%,P=0.003;3年OS率:92.3% vs 51.8%,P=0.040)。为排除年龄干扰,对 < 40岁的ALCL患者进行亚组分析,ALK+ALCL组的OS率仍显著优于ALK-ALCL组(P=0.002)。多因素分析显示,ALK表达为影响OS的独立良好预后因素。此外,ECOG评分和肺部受累为影响OS的独立不良预后因素。肺部受累的ALCL患者一线化疗CR率低(36.4%),且远期生存情况较差(中位OS为8个月,3年OS率为31.1%)。结论 ALCL整体较其他类型外周T细胞淋巴瘤预后相对良好。ALK+ALCL患者在化疗有效率和生存方面均显著优于ALK-ALCL患者。除ALK表达提示良好预后外,ECOG评分和肺部受累为影响ALCL患者OS的独立不良预后因素。在CHOP类化疗方案的基础上联合CD30单抗、组蛋白去乙酰化酶抑制剂等新药为目前外周T细胞淋巴瘤的治疗趋势。Abstract: Objective To evaluate the clinical characteristics, survival and prognostic factors of primary systemic anaplastic large cell lymphoma(ALCL).Methods Between January 2013 and March 2021, the clinical data of 48 patients with primary systemic ALCL who were treated in Peking Union Medical College Hospital were reviewed retrospectively.Results There were 31 males and 17 females, and the median age at diagnosis was 37(range, 18-79) years. 79.2% of patients were in Ann Arbor stage Ⅲ/Ⅳ and 68.8% had B symptoms. Extranodal involvement was relatively common with 54.2% of patients having more than 1 site of extranodal involvement. Of the 48 patients, 46 received initial chemotherapy. The first-line regimens included GDP-ML regimen in 6 patients, CHOP/CHOEP regimen in 27 patients, chidamide combined with CHOP/CHOEP regimen in 10 patients, and brentuximab vedotin(BV) combined with CHP regimen in 3 patients. The complete remission(CR) rate was 57.8% and the overall remission rate was 71.1%. The median follow-up time was 37(range, 1-101) months. The 1-year and 3-year progression-free survival rates were both 41.2% and the 1-year and 3-year overall survival(OS) rates were 71.1% and 63.2%, respectively. Despite younger onset age, patients with ALK+ALCL had similar clinical characteristics compared with patients with ALK-ALCL. Patients with ALK+ALCL showed significant higher CR rate and superior OS compared with patients with ALK-ALCL(the CR rates were 85.7% vs 41.2%,P=0.003, the 3-year OS rates were 92.3% vs 51.8%,P=0.040). To eliminate the confounding of age, subgroup analysis including only the ALCL patients under 40 years old showed that the OS of patients with ALK+ALCL was still significantly superior to that of ALK-ALCL(P=0.002). Multivariate analysis showed that ALK expression was an independent prognostic factor for ALCL. In addition, multivariate analysis showed that ECOG score and lung involvement were independent adverse prognostic factors for OS. The CR rate of ALCL patients with lung involvement was lower(36.4%) and the OS was poorer with a median OS 8 months and 3-year OS rate 31.1%.Conclusion In general, ALCL has a better prognosis than other types of peripheral T-cell lymphoma. Patients with ALK+ALCL showed a significant higher CR rate and superior OS compared with patients with ALK-ALCL. In addition to ALK expression which suggests good prognosis, ECOG score and lung involvement are independent adverse prognostic factors for OS in patients with ALCL. CHOP-like chemotherapies combined with new drugs such as BV and HDAC inhibitors are the current trend of peripheral T-cell lymphoma treatment.
-

-
表 1 ALK+与ALK-ALCL患者临床特点比较
例(%) 临床特征 ALK+
ALCL
(14例)ALK-
ALCL
(34例)χ2 P 中位年龄(范围)/岁 25
(13~66)50
(22~79)0.017 >60岁 1(7.1) 12(35.3) 5.730 0.046 存在B症状 11(78.6) 22(64.7) 1.454 0.228 Ann Arbor分期Ⅲ/Ⅳ 11(78.6) 27(79.4) 0.087 0.768 ECOG评分≥2分 4(28.6) 11(32.4) 0.011 0.917 LDH>250 U/L 4(28.6) 22(64.7) 5.636 0.022 结外受累器官>1个 7(50.0) 19(55.9) 0.022 0.883 IPI评分3~5分 6(42.9) 20(58.8) 1.052 0.305 一线化疗获得CR 12(85.7) 14(41.2) 4.741 0.003 表 2 影响ALCL患者OS的Cox回归多因素分析
因素 HR 95%CI P 年龄>60岁 0.562 ECOG评分 2.488
(每增加1分)1.636~3.782 < 0.001 ALK阳性 0.108 0.014~0.825 0.032 LDH>250 U/L 0.563 肺受累 3.370 1.118~10.155 0.031 胸膜受累 0.342 -
[1] Swerdlow SH, Campo E, Pileri SA, et al. The 2016 revision of the World Health Organization classification of lymphoid neoplasms[J]. Blood, 2016, 127(20): 2375-2390. doi: 10.1182/blood-2016-01-643569
[2] Sun J, Yang Q, Lu Z, et al. Distribution of lymphoid neoplasms in China: analysis of 4, 638 cases according to the World Health Organization classification[J]. Am J Clin Pathol, 2012, 138(3): 429-434. doi: 10.1309/AJCP7YLTQPUSDQ5C
[3] Cao C, Feng J, Gu H, et al. Distribution of lymphoid neoplasms in Northwest China: Analysis of 3244 cases according to WHO classification in a single institution[J]. Ann Diagn Pathol, 2018, 34: 60-65. doi: 10.1016/j.anndiagpath.2017.05.005
[4] Vose J, Armitage J, Weisenburger D. International T-Cell Lymphoma Project. International peripheral T-cell and natural killer/T-cell lymphoma study: pathology findings and clinical outcomes[J]. J Clin Oncol, 2008, 26(25): 4124-4130. doi: 10.1200/JCO.2008.16.4558
[5] International Non-Hodgkin's Lymphoma Prognostic Factors Project. A predictive model for aggressive non-Hodgkin's lymphoma[J]. N Engl J Med, 1993, 329(14): 987-994. doi: 10.1056/NEJM199309303291402
[6] Gallamini A, Stelitano C, Calvi R, et al. Peripheral T-cell lymphoma unspecified(PTCL-U): a new prognostic model from a retrospective multicentric clinical study[J]. Blood, 2004, 103(7): 2474-2479. doi: 10.1182/blood-2003-09-3080
[7] Zhang Y, Zhang W, Li J, et al. Gemcitabine, cisplatin, and dexamethasone(GDP)in combination with methotrexate and pegaspargase is active in newly diagnosed peripheral T cell lymphoma patients: a phase 2, single-center, open-label study in China[J]. Ann Hematol, 2019, 98(1): 143-150. doi: 10.1007/s00277-018-3488-1
[8] Cheson BD, Fisher RI, Barrington SF, et al. Recommendations for Initial Evaluation, Staging, and Response Assessment of Hodgkin and non-Hodgkin Lymphoma: The Lugano Classification[J]. J Clin Onclol, 2014, 32(27): 3059-3068. doi: 10.1200/JCO.2013.54.8800
[9] Tsuyama N, Sakamoto K, Sakata S, et al. Anaplastic large cell lymphoma: pathology, genetics, and clinical aspects[J]. J Clin Exp Hematop, 2017, 57(3): 120-142. doi: 10.3960/jslrt.17023
[10] Savage KJ, Harris NL, Vose JM, et al. ALK- anaplastic large-cell lymphoma is clinically and immunophenotypically different from both ALK+ ALCL and peripheral T-cell lymphoma, not otherwise specified: report from the International Peripheral T-Cell Lymphoma Project[J]. Blood, 2008, 111(12): 5496-5504. doi: 10.1182/blood-2008-01-134270
[11] Sibon D, Fournier M, Brière J, et al. Long-term outcome of adults with systemic anaplastic large-cell lymphoma treated within the Groupe d'Etude des Lymphomes de l'Adulte trials[J]. J Clin Oncol, 2012, 30(32): 3939-3946. doi: 10.1200/JCO.2012.42.2345
[12] Armitage JO. The aggressive peripheral T-cell lymphomas: 2017[J]. Am J Hematol, 2017, 92(7): 706-715. doi: 10.1002/ajh.24791
[13] Liu W, Ji X, Song Y, et al. Improving survival of 3760 patients with lymphoma: Experience of an academic center over two decades[J]. Cancer Med, 2020, 9(11): 3765-3774. doi: 10.1002/cam4.3037
[14] 陈英坤, 俞文娟, 刘辉, 等. 原发系统型间变性大细胞淋巴瘤40例临床特征及预后分析[J]. 中华血液学杂志, 2020, 41(3): 222-227.
[15] Parrilla Castellar ER, Jaffe ES, Said JW, et al. ALK-negative anaplastic large cell lymphoma is a genetically heterogeneous disease with widely disparate clinical outcomes[J]. Blood, 2014, 124(9): 1473-1480. doi: 10.1182/blood-2014-04-571091
[16] Schmitz N, Trümper L, Ziepert M, et al. Treatment and prognosis of mature T-cell and NK-cell lymphoma: an analysis of patients with T-cell lymphoma treated in studies of the German High-Grade Non-Hodgkin Lymphoma Study Group[J]. Blood, 2010, 116(18): 3418-3425. doi: 10.1182/blood-2010-02-270785
[17] Horwitz S, O'Connor OA, Pro B, et al. Brentuximab vedotin with chemotherapy for CD30-positive peripheral T-cell lymphoma(ECHELON-2): a global, double-blind, randomised, phase 3 trial[J]. Lancet, 2019, 393(10168): 229-240. doi: 10.1016/S0140-6736(18)32984-2
[18] Shi Y, Jia B, Xu W, et al. Chidamide in relapsed or refractory peripheral T cell lymphoma: a multicenter real-world study in China[J]. J Hematol Oncol, 2017, 10(1): 69. doi: 10.1186/s13045-017-0439-6
[19] Park SI, Horwitz SM, Foss FM, et al. COMPLETE Investigators. The role of autologous stem cell transplantation in patients with nodal peripheral T-cell lymphomas in first complete remission: Report from COMPLETE, a prospective, multicenter cohort study[J]. Cancer, 2019, 125(9): 1507-1517. doi: 10.1002/cncr.31861
[20] 卢柠, 李晓帆, 董玉君, 等. 造血干细胞移植治疗间变大细胞淋巴瘤的临床分析[J]. 中华血液学杂志, 2020, 41(2): 117-122.
-




 下载:
下载: