Analysis of erythroid lineage features in myelodysplastic syndromes patients with SF3B1 gene mutation
-
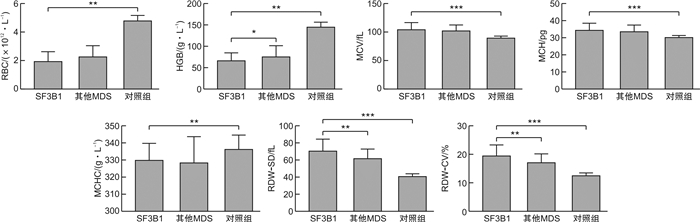
摘要: 目的 观察剪接因子3B亚单位1(SF3B1)基因突变的骨髓增生异常综合征(MDS)患者红系造血特征。方法 以2018年8月至2021年11月初诊126例MDS患者为研究对象,根据是否存在SF3B1基因突变分为SF3B1突变组与SF3B1野生组,比较2组之间红细胞参数:外周血中红细胞(RBC)、血红蛋白(HGB)、平均红细胞体积(MCV)、平均红细胞血红蛋白量(MCH)、平均红细胞血红蛋白浓度(MCHC)、红细胞分布宽度标准差(RDW-SD)、红细胞分布宽度变异系数(RDW-CV)、骨髓中红系比例、粒红比的差别,并比较SF3B1突变组与同期100例健康体检者(对照组)外周血各参数的差别。分析SF3B1突变组与SF3B1野生组HGB与MCV、RDW-SD、RDW-CV的相关性。结果 ① SF3B1突变组HGB低于SF3B1野生组(P< 0.05),而RDW-SD、RDW-CV高于SF3B1野生组(P< 0.01),2组RBC、MCV、MCH、MCHC比较差异无统计学意义(P>0.05)。SF3B1突变组RBC、HGB、MCHC均明显低于对照组(P< 0.01),而MCV、MCH、RDW-SD、RDW-CV均明显高于对照组(P< 0.001)。②SF3B1突变组与SF3B1野生组骨髓中红系比例无明显差异[(39.808 0±17.636 4)% vs(33.800 0±18.281 3)%,P=0.135],粒红比无明显差异[1.02(0.04~4.29) vs 1.14(0.11~154),P=0.615]。③SF3B1突变组HGB与MCV、RDW-SD、RDW-CV均无关,SF3B1野生组HGB与MCV无关,与RDW-SD、RDW-CV呈负相关。结论 SF3B1基因突变的MDS患者红细胞存在更高的异质性;无SF3B1基因突变的MDS患者HGB水平与红细胞变异程度呈负相关。
-
关键词:
- 剪接因子3B亚单位1 /
- 骨髓增生异常综合征 /
- 红细胞
Abstract: Objective To investigate the erythroid lineage features in myelodysplastic syndromes(MDS) patients with SF3B1 gene mutation.Methods One hundred and twenty-six newly diagnosed MDS patients between August 2018 and November 2021 were enrolled. They were divided into two groups, mutational SF3B1 group and wild-type SF3B1 group. The red blood cell(RBC) related parameters including RBC, hemoglobin(HGB), mean corpuscular volume(MCV), mean corpuscular hemoglobin(MCH), mean corpuscu larhemoglobin concentration(MCHC), standard deviation of red blood cell distribution width(RDW-SD), coefficient of variation of red blood cell distribution width(RDW-CV) in peripheral blood(PB), erythroid percentage and G/E ratio in bone marrow(BM) were compared between two groups. PB parameters were also compared between mutational SF3B1 group and one hundred healthy controls(HC). The relationships between HGB and MCV, RDW-SD, RDW-CV were analyzed in mutational and wild-type SF3B1 groups respectively.Results ① Compared with wild-type SF3B1 group, mutational SF3B1 group had lower HGB(P< 0.05), higher RDW-SD, RDW-CV(P< 0.01). There was no significant difference in RBC, MCV, MCH, MCHC between two groups. Compared with HC, mutational SF3B1 group had lower RBC, HGB, MCHC(P< 0.01) and higher MCV, MCH, RDW-SD, RDW-CV(P< 0.001). ②Erythroid percentage was not significantly different between mutational and wild-type SF3B1 groups([39.808 0±17.636 4]% vs[33.800 0±18.281 3]%,P=0.135). G/E ratio was neither significantly different between these two groups(1.02[0.04-4.29]vs 1.14[0.11-154],P=0.615). ③In mutational SF3B1 group, HGB had no relationships with MCV, RDW-SD, RDW-CV. In wild-type SF3B1 group, HGB was negatively correlated with RDW-SD and RDW-CV while not significantly correlated with MCV.Conclusion RBC showed larger heterogeneity in MDS patients with SF3B1 mutation than without. HGB may be negatively correlated with RBC diversity in MDS patients without SF3B1 mutation.-
Key words:
- splicing factor 3B subunit 1 /
- myelodysplastic syndromes /
- red blood cell
-

-
表 1 SF3B1突变组HGB与MCV、RDW-SD、RDW-CV的相关性
参数 MCV RDW-SD RDW-CV r -0.128 -0.160 -0.165 P 0.544 0.445 0.432 表 2 SF3B1野生组HGB与MCV、RDW-SD、RDW-CV的相关性
参数 MCV RDW-SD RDW-CV r -0.082 -0.261 -0.280 P 0.415 0.009 0.004 -
[1] 龚婧晔, 肖志坚. 骨髓增生异常综合征患者感染危险因素及预防[J]. 临床血液学杂志, 2021, 34(1): 65-70. https://www.cnki.com.cn/Article/CJFDTOTAL-LCXZ202101016.htm
[2] 蔡亚楠, 徐泽锋, 李冰, 等. 伴环状铁粒幼红细胞增多骨髓增生异常综合征基因突变特征及临床意义[J]. 中华血液学杂志, 2020, 41(5): 379-386. https://cdmd.cnki.com.cn/Article/CDMD-10023-1021026344.htm
[3] Venable ER, Chen D, Chen CP, et al. Pathologic Spectrum and Molecular Landscape of Myeloid Disorders Harboring SF3B1 Mutations[J]. Am J Clin Pathol, 2021, 156(4): 679-690. doi: 10.1093/ajcp/aqab010
[4] Xiong B, Xue M, Yu Y, et al. SF3B1 Mutation but Not Ring Sideroblasts Identifies a Specific Group of Myelodysplastic Syndrome-Refractory Cytopenia With Multilineage Dysplasia[J]. Clin Lymphoma Myeloma Leuk, 2020, 20(5): 329-339. doi: 10.1016/j.clml.2020.01.005
[5] Yan X, Wang L, Jiang L, et al. Clinical significance of cytogenetic and molecular genetic abnormalities in 634 Chinese patients with myelodysplastic syndromes[J]. Cancer Med, 2021, 10(5): 1759-1771. doi: 10.1002/cam4.3786
[6] Malcovati L, Stevenson K, Papaemmanuil E, et al. SF3B1-mutant MDS as a distinct disease subtype: a proposal from the International Working Group for the Prognosis of MDS. Blood. 2020;136(2): 157-170[J]. Blood, 2021, 137(21): 3003. doi: 10.1182/blood.2021012067
[7] Duetz C, Westers TM, In't Hout F, et al. Distinct bone marrow immunophenotypic features define the splicing factor 3B subunit 1(SF3B1)-mutant myelodysplastic syndromes subtype[J]. Br J Haematol, 2021, 193(4): 798-803. doi: 10.1111/bjh.17414
[8] 骨髓增生异常综合征中国诊断与治疗指南(2019年版)[J]. 中华血液学杂志, 2019, 40(2): 89-97.
[9] Bastida JM, López-Godino O, Vicente-Sánchez A, et al. Hidden myelodysplastic syndrome(MDS): A prospective study to confirm or exclude MDS in patients with anemia of uncertain etiology[J]. Int J Lab Hematol, 2019, 41(1): 109-117. doi: 10.1111/ijlh.12933
[10] 宋晓颖, 袁宝军, 崔晶晶, 等. 骨髓增生异常综合征患者红细胞参数水平变化及临床意义[J]. 国际检验医学杂志, 2020, 41(13): 1629-1631, 1635. doi: 10.3969/j.issn.1673-4130.2020.13.022
[11] 史仲珣, 秦铁军, 徐泽锋, 等. 平均红细胞体积≤100 fl是骨髓原始细胞 < 5%骨髓增生异常综合征患者的独立预后因素[J]. 中华血液学杂志, 2020, 41(1): 28-33.
[12] Shingai N, Harada Y, Iizuka H, et al. Impact of splicing factor mutations on clinical features in patients with myelodysplastic syndromes[J]. Int J Hematol, 2018, 108(6): 598-606. doi: 10.1007/s12185-018-2551-y
[13] 蔡亚楠, 徐泽锋, 李冰, 等. 伴环状铁粒幼红细胞增多骨髓增生异常综合征基因突变特征及临床意义[J]. 中华血液学杂志, 2020, 41(5): 379-386. https://cdmd.cnki.com.cn/Article/CDMD-10023-1021026344.htm
[14] Obeng EA, Chappell RJ, Seiler M, et al. Physiologic Expression of Sf3b1(K700E)Causes Impaired Erythropoiesis, Aberrant Splicing, and Sensitivity to Therapeutic Spliceosome Modulation[J]. Cancer Cell, 2016, 30(3): 404-417. doi: 10.1016/j.ccell.2016.08.006
[15] Baba Y, Saito B, Shimada S, et al. Association of red cell distribution width with clinical outcomes in myelodysplastic syndrome[J]. Leuk Res, 2018, 67: 56-59. doi: 10.1016/j.leukres.2018.02.004
[16] Mortera-Blanco T, Dimitriou M, Woll PS, et al. SF3B1-initiating mutations in MDS-RSs target lymphomyeloid hematopoietic stem cells[J]. Blood, 2017, 130(7): 881-890. doi: 10.1182/blood-2017-03-776070
[17] Dolatshad H, Pellagatti A, Fernandez-Mercado M, et al. Disruption of SF3B1 results in deregulated expression and splicing of key genes and pathways in myelodysplastic syndrome hematopoietic stem and progenitor cells[J]. Leukemia, 2015, 29(8): 1798. doi: 10.1038/leu.2015.178
[18] De La Garza A, Cameron RC, Gupta V, et al. The splicing factor Sf3b1 regulates erythroid maturation and proliferation via TGFβ signaling in zebrafish[J]. Blood Adv, 2019, 3(14): 2093-2104. doi: 10.1182/bloodadvances.2018027714
[19] Huang Y, Hale J, Wang Y, et al. SF3B1 deficiency impairs human erythropoiesis via activation of p53 pathway: implications for understanding of ineffective erythropoiesis in MDS[J]. J Hematol Oncol, 2018, 11(1): 19. doi: 10.1186/s13045-018-0558-8
[20] Lieu YK, Liu Z, Ali AM, et al. SF3B1 mutant-induced missplicing of MAP3K7 causes anemia in myelodysplastic syndromes[J]. Proc Natl Acad Sci U S A, 2022, 119(1): e2111703119. doi: 10.1073/pnas.2111703119
[21] Clough CA, Pangallo J, Sarchi M, et al. Coordinated missplicing of TMEM14C and ABCB7 causes ring sideroblast formation in SF3B1-mutant myelodysplastic syndrome[J]. Blood. 2022, 139(13): 2038-2049. doi: 10.1182/blood.2021012652
-




 下载:
下载: