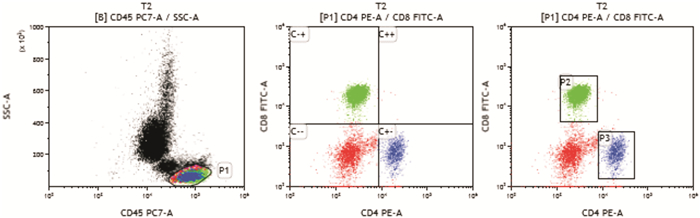
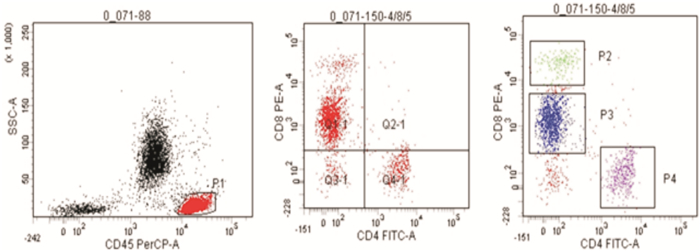
-
摘要: 目的 检测EB病毒(EBV)感染所致成人传染性单核细胞增多症(IM)患者外周血淋巴细胞亚群和血浆EBV DNA载量,分析EBV载量与淋巴细胞亚群的相关性。方法 收集2016至2021年确诊的成人IM患者318例作为IM组,选择同期体检的健康成人31例作为对照组。采用多参数流式细胞术检测外周血各淋巴细胞亚群占总淋巴细胞百分比,实时荧光定量PCR测量血浆中的EBV DNA载量。结果 IM组CD8+CD3+(53.39±8.02)%和CD8弱阳性CD3+(12.17±8.52)%比例明显增高,与对照组比较差异有统计学意义(P< 0.001);CD4+CD3+(30.7±9.83)%和CD4+/CD8+(0.60±0.25)明显降低,与对照组比较差异有统计学意义(P< 0.05);IM组CD3+、CD16+CD56+CD3-比例和对照组比较差异均无统计学意义(P>0.05);IM组EBV DNA载量与CD8+CD3+和CD8弱阳性CD3+比例呈正相关(P< 0.05),与CD3+、CD4+CD3+、CD16+CD56+CD3-比例无相关性(P>0.05)。结论 成人IM患者外周血CD8+T淋巴细胞和CD8弱阳性T淋巴细胞显著增多,EBV DNA载量与CD8+T淋巴细胞和CD8弱阳性T淋巴细胞呈正相关,综合淋巴细胞亚群检测结果与血浆EBV DNA载量有助于疾病的诊断。
-
关键词:
- EB病毒 /
- 传染性单核细胞增多症 /
- 淋巴细胞亚群
Abstract: Objective To detect the peripheral blood lymphocyte subsets and plasma EBV DNA load in adult infectious mononucleosis patients infected by EBV virus, and analyze the correlation between EB virus load and lymphocyte subsets.Methods A total of 318 adult infectious mononucleosis patients diagnosed in our hospital from 2016 to 2021 were collected as the experimental group(IM group), and 31 healthy adults who underwent physical examination during the same period were selected as the control group. Multi-parameter flow cytometry was used to detect the percentage of lymphocyte subsets in peripheral blood to total lymphocytes, and real-time fluorescence quantitative PCR was used to measure EBV DNA load in plasma.Results The proportion of CD8+CD3+(53.39±8.02)% and CD8 weakly positive CD3+(12.17±8.52)% in IM group were significantly higher than those in control group(P< 0.001). CD4+CD3+(30.7±9.83)% and CD4+/CD8+(0.60±0.25) were significantly lower than those in the control group(P< 0.05). There was no significant difference in the ratio of IM CD3+, CD16+CD56+CD3- between IM group and control group. In IM group, EBV DNA load was positively correlated with the proportion of CD8+CD3+ and CD8 weakly positive CD3+(P< 0.05), but not correlated with the proportion of CD3+, CD4+CD3+and CD16+CD56+CD3-(P>0.05).Conclusion CD8+T lymphocytes and CD8 weakly positive T lymphocytes in peripheral blood of adult IM patients significantly increased, and EBV DNA load was positively correlated with the proportion of CD8+T lymphocytes and CD8 weakly positive T lymphocytes. Comprehensive detection results of lymphocyte subsets and plasma EBV DNA load may be helpful for diagnosis of the disease.-
Key words:
- Epstein-Barr virus /
- infectious mononucleosis /
- lymphocyte subsets
-

-
表 1 IM组与对照组淋巴细胞亚群比较分析
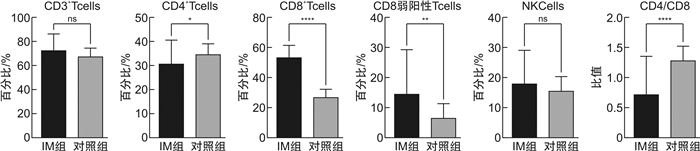
X ±S 组别 CD3+/% CD4+CD3+/% CD8+CD3+/% CD8弱阳性CD3+/% CD16+CD56+CD3-/% CD4+/CD8+ IM组 72.66±13.58 30.7±9.83 53.39±8.02 12.17±8.52 18.21±10.98 0.60±0.25 对照组 68.13±6.43 34.67±4.30 27.36±4.81 6.58±4.66 15.79±4.70 1.30±0.23 t 1.83 -2.22 17.75 3.60 1.22 -14.87 P 0.07 0.03 < 0.001 < 0.001 0.230 < 0.001 表 2 IM组患者EBV DNA载量与淋巴细胞亚群比例相关性分析
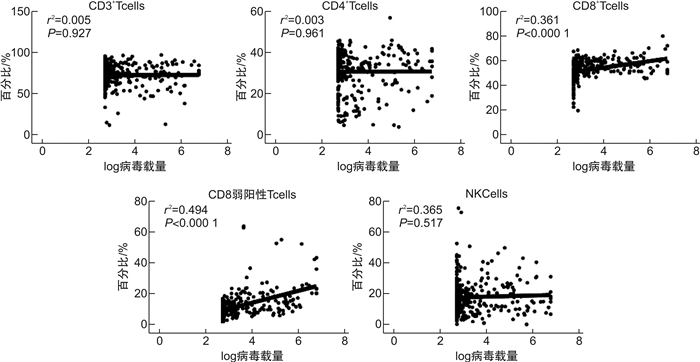
比值 CD3+/% CD4+CD3+/% CD8+CD3+/% CD8弱阳性CD3+/% CD16+CD56+CD3-/% R 0.043 0.029 0.161 0.378 -0.012 P 0.446 0.606 0.004 < 0.001 0.832 -
[1] 胡岩岩, 潘家华, 周浩泉. 儿童传染性单核细胞增多症临床及实验室检查特点分析[J]. 中华全科医学, 2021, 19(9): 1510-1513. https://www.cnki.com.cn/Article/CJFDTOTAL-SYQY202109022.htm
[2] Womack J, Jimenez M. Common questions about infectious mononucleosis[J]. Am Fam Physician, 2015, 91(6): 372-376.
[3] Tangye SG, Palendira U, Edwards ES. Human immunity against EBV-lessons from the clinic[J]. J Exp Med, 2017, 214(2): 269-283. doi: 10.1084/jem.20161846
[4] Sulik A, Oldak E, Kroten A, et al. Epstein-Barr virus effect on frequency of functionally distinct T cell subsets in children with infectious mononucleosis[J]. Adv Med Sci, 2014, 59(2): 227-231. doi: 10.1016/j.advms.2014.04.003
[5] 景清. 小儿传染性单核细胞增多症112例临床分析[J]. 四川医学, 2018, 39(1): 79-82. https://www.cnki.com.cn/Article/CJFDTOTAL-SCYX201801024.htm
[6] 荣婷婷, 王维维, 王娟娟, 等. EB病毒感染患儿淋巴细胞亚群特征和EBV DNA载量分析[J]. 检验医学, 2018, 33(4): 285-289. doi: 10.3969/j.issn.1673-8640.2018.04.003
[7] 孙敏, 盛艳蕊, 周洪静. 成人传染性单核细胞增多症临床表现及实验室检查特点分析[J]. 中国药物与临床, 2020, 20(10): 1707-1708. https://www.cnki.com.cn/Article/CJFDTOTAL-YWLC202010063.htm
[8] Chatterjee B, Deng Y, Holler A, et al. CD8+T cells retain protective functions despite sustained inhibitory receptor expression during Epstein-Barr virus infection in vivo[J]. PLoS Pathog, 2019, 15(5): e1007748. doi: 10.1371/journal.ppat.1007748
[9] Maini MK, Gudgeon N, Wedderburn LR, et al. Clonal expansions in acute EBV infection are detectable in the CD8 and not the CD4 subset and persist with a variable CD45 phenotype[J]. J Immunol, 2000, 165(10): 5729-5737. doi: 10.4049/jimmunol.165.10.5729
[10] Hoshino Y, Morishima T, Kimura H, et al. Antigen-driven expansion and contraction of CD8+-activated T cells in primary EBV infection[J]. J Immunol, 1999, 163(10): 5735-5740.
[11] 谢静, 王焕玲, 邱志峰, 等. 成人传染性单核细胞增多症和慢性活动性EB病毒感染外周血淋巴细胞亚群分析[J]. 中华内科杂志, 2016, 55(6): 455-459. doi: 10.3760/cma.j.issn.0578-1426.2016.06.011
[12] Chen T, Chen Y, Bao W, et al. T-lymphocyte subsets and Th1/Th2 cytokines in convalescent patients with Epstein-Barr virus-associated aplastic anemia[J]. Hematology, 2020, 25(1): 11-16. doi: 10.1080/16078454.2019.1702304
[13] Song YL, Wang BF, Jiang NG, et al. CD8dimCD3+ lymphocytes in fever patients might be biomarkers of active EBV infection and exclusion indicator of T-LGLL[J]. Biomark Med, 2020, 14(18): 1703-1715. doi: 10.2217/bmm-2020-0456
[14] Taylor GS, Long HM, Brooks JM, et al. The immunology of Epstein-Barr virus-induced disease[J]. Annu Rev Immunol, 2015, 33: 787-821. doi: 10.1146/annurev-immunol-032414-112326
[15] Balfour HH Jr, Holman CJ, Hokanson KM, et al. A prospective clinical study of Epstein-Barr virus and host interactions during acute infectious mononucleosis[J]. J Infect Dis, 2005, 192(9): 1505-1512. doi: 10.1086/491740
[16] Wingate PJ, McAulay KA, Anthony IC, et al. Regulatory T cell activity in primary and persistent Epstein-Barr virus infection[J]. J Med Virol, 2009, 81(5): 870-877. doi: 10.1002/jmv.21445
-

| 引用本文: | 卢峰, 李甫罡, 罗志刚, 等. CD8+T淋巴细胞在成人传染性单核细胞增多症患者中的表达分析[J]. 临床血液学杂志, 2022, 35(6): 423-427. doi: 10.13201/j.issn.1004-2806.2022.06.008 |
| Citation: | LU Feng, LI Fugang, LUO Zhigang, et al. Analysis of expression of CD8+T lymphocytes in adult infectious mononucleosis[J]. J Clin Hematol, 2022, 35(6): 423-427. doi: 10.13201/j.issn.1004-2806.2022.06.008 |
- Figure 1.
- Figure 2.
- Figure 3.
- Figure 4.



 下载:
下载: