Value of HPA gene polymorphism in evaluating efficacy of platelet transfusion in patients with liver disease
-
摘要: 目的 探究血小板抗原(HPA)基因多态性对肝病患者血小板输注疗效的评估价值及血小板输注无效(PTR)的影响因素。方法 以2019年1月至2021年7月治疗的98例肝病患者为研究对象,根据PTR发生情况分为PTR组(25例)及对照组(73例),采用问卷调查收集患者年龄、性别、原发疾病、是否合并感染、是否存在发热症状等临床资料。应用德国QIAGEN公司血液DNA提取试剂盒提取总DNA,行HPA基因多态性检测。结果 本研究纳入肝病患者的HPA-2、HPA-3、HPA-15基因多态性均符合Hardy-Weinberg平衡(P>0.05);PTR组HPA-2、HPA-3、HPA-15基因ab、bb型检出率均高于对照组(P< 0.05);HPA-2、HPA-3、HPA-15基因多态性联合检测评估肝病患者血小板输注疗效的ROC曲线下面积(AUC)>0.75;PTR组在存在发热症状、合并感染、血小板体外保存时间>3 d、输注次数≥3次、脾脏厚度≥40 mm的人数比例高于对照组(P< 0.05),均为影响肝病患者出现PTR的危险因素(P< 0.05)。结论 HPA基因多态性对肝病患者血小板输注疗效具有评估价值,且发热、合并感染、血小板体外保存时间>3 d、输注次数≥3次、脾脏厚度≥40 mm是影响肝病患者出现PTR的危险因素。Abstract: Objective To explore the evaluation value of polymorphisms of platelet antigen(HPA) gene for platelet transfusion effect and the influencing factors of platelet transfusion refractoriness(PTR) in hepatopathy patients.Methods A total of 98 patients with liver disease from January 2019 to July 2021 were divided into PTR group(25 cases) and control group(73 cases) according to the occurrence of PTR. The clinical data such as age, gender, primary disease, CO infection and fever symptoms were investigated by questionnaire. The total DNA was extracted with the blood DNA extraction kit of Qiagen company in Germany, and the HPA gene polymorphism was detected.Results The polymorphisms of HPA-2, HPA-3 and HPA-15 genes were all in accordance with Hardy-Weinberg equilibrium(P>0.05). The detection rates of types ab and bb in HPA-2, HPA-3 and HPA-15 genes in PTR group were higher than those in control group(P< 0.05). AUC of HPA-2 combined with HPA-3 and HPA-15 polymorphisms for evaluating platelet transfusion effect was over 0.75. The proportions of cases with fever, infection, platelet in vitro storage time>3 d, transfusion times ≥3 times and spleen thickness ≥40 mm in PTR group were higher than those in control group(P< 0.05). The fever, infection, platelet in vitro storage time>3 d, transfusion times ≥3 times and spleen thickness ≥40 mm were risk factors of PTR(P< 0.05).Conclusion There may be evaluation value of HPA gene polymorphisms for platelet transfusion effect in hepatopathy patients. Fever, infection, platelet in vitro storage time>3 d, transfusion times ≥3 times and spleen thickness ≥40 mm might be risk factors of PTR.
-
Key words:
- hepatopathy /
- platelet antigen /
- platelet transfusion effect
-

-
表 1 患者HPA基因的Hardy-Weinberg平衡分析
频率 HPA-2 HPA-3 HPA-15 aa ab bb aa ab bb aa ab bb 实际频率 39.00 37.00 22.00 32.00 41.00 25.00 29.00 38.00 31.00 理论频率 33.74 47.53 16.74 28.13 48.75 21.13 23.51 48.98 25.51 χ2 2.406 1.245 2.493 P 0.300 0.537 0.288 表 2 PTR组及对照组的HPA基因多态性比较
例 组别 例数 HPA-2 HPA-3 HPA-15 aa ab bb aa ab bb aa ab bb PTR组 25 7 15 3 4 16 5 4 18 3 对照组 73 32 22 19 28 25 20 25 20 28 χ2 7.204 7.190 15.739 P 0.027 0.028 < 0.001 表 3 HPA基因多态性对肝病患者血小板输注疗效的评估价值分析
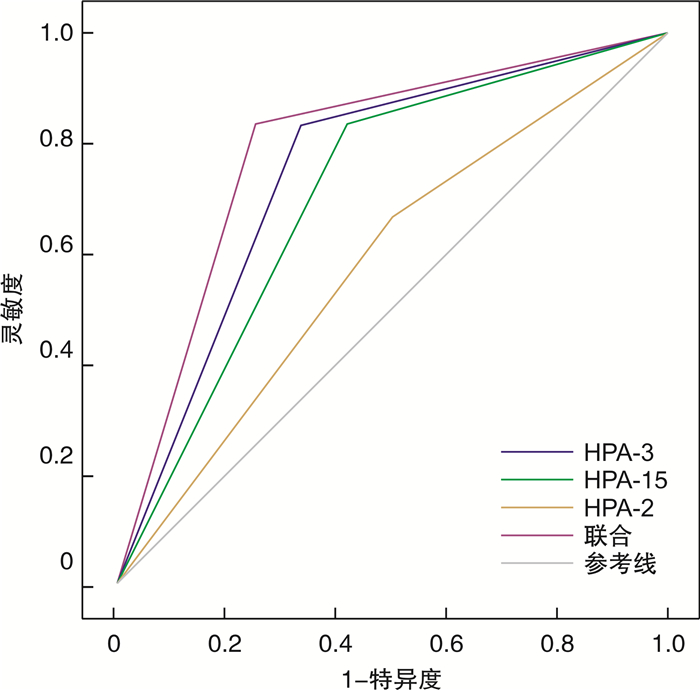
因素 AUC SE 95% CI 特异度/% 敏感度/% HPA-2 0.596 0.065 0.470~0.723 72.00 47.22 HPA-3 0.726 0.056 0.615~0.836 84.00 61.11 HPA-15 0.684 0.059 0.569~0.799 84.00 52.78 联合检测 0.791 0.05 0.694~0.888 84.00 63.89 表 4 影响肝病患者出现PTR的单因素分析
例(%) 因素 PTR组(25例) 对照组(73例) χ2 P 性别 3.124 0.077 男 15(60.00) 57(78.08) 女 10(40.00) 16(21.92) 年龄 0.908 0.341 ≥60岁 9(36.00) 19(26.03) < 60岁 16(64.00) 54(73.97) 发热 8.663 0.003 是 17(68.00) 25(34.25) 否 8(32.00) 48(65.75) 疾病类型 0.523 0.971 慢性肝炎 10(40.00) 27(36.99) 重症肝炎 6(24.00) 19(26.03) 肝硬化 5(20.00) 12(16.44) 肝癌 2(8.00) 9(12.33) 肝衰竭 2(8.00) 6(8.22) 合并感染 14.117 < 0.001 是 17(68.00) 19(26.03) 否 8(32.00) 54(73.97) 血小板体外保存时间 12.012 < 0.001 ≤3 d 15(60.00) 66(90.41) > 3 d 10(40.00) 7(9.59) 输注次数 4.859 0.028 ≥3次 17(68.00) 31(42.47) < 3次 8(32.00) 42(57.53) 脾脏厚度 4.027 0.045 ≥40 mm 15(60.00) 27(36.99) < 40 mm 10(40.00) 46(63.01) 活动性出血 2.832 0.092 是 11(44.00) 19(26.03) 否 14(56.00) 54(73.97) 表 5 影响肝病患者出现PTR的多因素分析
因素 β SE Wald χ2 OR 95% CI P 发热 0.597 0.301 3.934 1.817 1.007~3.277 0.048 合并感染 0.657 0.321 4.189 1.929 1.028~3.619 0.041 血小板体外保存时间 0.618 0.286 4.669 1.855 1.059~3.250 0.031 输注次数 0.738 0.354 4.346 2.092 1.045~4.186 0.038 脾脏厚度 0.692 0.279 6.152 1.998 1.156~3.452 0.014 赋值:PTR(是为1,否为0);发热(是为1,否为0);合并感染(是为1,否为0);血小板体外保存时间(> 3 d为1, < 3 d为0);输注次数(≥3次为1, < 3次为0);脾脏厚度(≥40 mm为1, < 40 mm为0)。 -
[1] Arnone GD, Kumar P, Wonais MC, et al. Impact of Platelet Transfusion on Intracerebral Hemorrhage(ICH)in Patients on Antiplatelet Therapy-An Analysis Based on ICH Score[J]. World Neurosurg, 2018, 111(5): e895-e904.
[2] 张桂芬, 闫萌. 反复输血后血小板输注无效患者抗体阳性率、特异性及影响因素分析[J]. 临床血液学杂志, 2020, 33(6): 391-393. https://www.cnki.com.cn/Article/CJFDTOTAL-LCXZ202006004.htm
[3] Mangerona CM, Garcia FB, Moraes-Souza H. Frequency of human platelet antigens(HPA)-1, -2, -5 and-15 in Brazilian blood donors and establishment of a panel of HPA-typed donors[J]. Transfus Med, 2015, 25(3): 189-194. doi: 10.1111/tme.12210
[4] Bertrand G, Aarnink A, Nivet C, et al. Le cas d'un génotypage HPA-3 erroné en raison de la présence d'un antigène rare HPA-27bw chez un nouveau-né sévèrement thrombopénique[J]. Transfus Clin Biol, 2017, 24(3): 342-343.
[5] 胡丽华. 临床输血检验[M]. 北京: 中国医药科技出版社, 2004: 118-119, 274-275.
[6] 池肇春. 实用临床肝病学[M]. 北京: 人民军医出版社, 2015: 25-26, 31-35.
[7] Bal Dit Sollier C, Berge N, Hamadouche S, et al. Is platelet function testing at the acute phase under P2Y12 inhibitors helpful in predicting bleeding in real-life patients with acute coronary syndrome? The AVALANCHE study[J]. Arch Cardiovasc Dis, 2021, 114(10): 612-623. doi: 10.1016/j.acvd.2021.06.003
[8] 钟丽红, 吴泳彬, 杨定坤, 等. 血小板输注无效的原因及其配型疗效的相关研究[J]. 血栓与止血学, 2018, 24(2): 53-56. https://www.cnki.com.cn/Article/CJFDTOTAL-XSZX201802015.htm
[9] 李玉梅, 徐恒仕, 王志良. HPA基因多态性及其等位基因对血小板输注效果的影响[J]. 中国输血杂志, 2017, 30(7): 744-747. https://www.cnki.com.cn/Article/CJFDTOTAL-BLOO201707038.htm
[10] Sut C, Tariket S, Cognasse F, et al. Determination of predictors of severity for recipient adverse reactions during platelet product transfusions[J]. Transfus Clin Biol, 2017, 24(2): 87-91. doi: 10.1016/j.tracli.2017.04.002
[11] 龚道方, 龙海波, 刘昭国, 等. 临床276例次单采血小板输注效果及相关因素分析[J]. 临床输血与检验, 2018, 20(4): 365-368. doi: 10.3969/j.issn.1671-2587.2018.04.009
[12] 徐朝霞, 杨妞, 罗佳. 163例血小板输注无效分析及血小板基因库应用[J]. 中国医师杂志, 2017, 19(12): 1884-1885. doi: 10.3760/cma.j.issn.1008-1372.2017.12.040
[13] Zhi H, Ahlen MT, Thinn AMM, et al. Atomic-Level Dissection of the Polyclonal Immune Response to the Human Platelet Alloantigen, HPA-1a(PlA1)[J]. Blood, 2018, 132(21): 3001-3011.
[14] Wienzek-Lischka S, König IR, Papenkort EM, et al. HLA-DRB3*01: 01 is a predictor of immunization against human platelet antigen-1a but not of the severity of fetal and neonatal alloimmune thrombocytopenia[J]. Transfusion, 2017, 57(3): 533-540. doi: 10.1111/trf.13950
[15] Seike K, Fujii N, Asano N, et al. Efficacy of HLA virtual cross-matched platelet transfusions for platelet transfusion refractoriness in hematopoietic stem cell transplantation[J]. Transfusion, 2020, 60(3): 473-478. doi: 10.1111/trf.15664
-




 下载:
下载: