Efficacy of a new induction regimen in the treatment of early T-cell precursor acute lymphoblastic leukemia
-
摘要: 目的 成人急性淋巴细胞白血病尚无标准诱导治疗方案,急性早前T淋巴细胞白血病(ETP-ALL)对标准的强化化疗反应差,复发风险高,预后差。拟探讨合适的诱导方案来提高其缓解率,延长患者生存期。方法 回顾性分析2017年1月—2020年12月接受不同方案诱导化疗的21例初诊ETP-ALL患者的疗效及主要不良反应。初诊ETP-ALL根据骨髓细胞学、流式细胞术免疫表型检测结果诊断。结果 21例患者中男14例,女7例;中位年龄27(16~57)岁;7例接受PCIOD方案[培门冬酶注射液:2500 IU/m2肌肉注射,第1、14天;环磷酰胺:800 mg·m-2·d-1,第1天;伊达比星:6~8 mg(总量10 mg),第1~3天;长春地辛:4 mg,第1天;地塞米松:20 mg/d,第1~7天]治疗。14例接受VTLP样方案(吡柔比星:40 mg/m2分3 d或柔红霉素30~45 mg·m-2·d-1,第8、15、22、29天;长春地辛:3 mg/m2或长春新碱1.5 mg/m2,第8、15、22、29天;培门冬酶注射液:2500 IU /m2,第14、28天;或左旋门冬酰胺酶5000 U /m2,第8、11、14、17、20、23、26、29天;±环磷酰胺:800 mg·m-2·d-1,第1天;泼尼松1 mg·kg-1·d-1,第1~14天,后改为口服逐渐减量到第30天停用,每28 d为一个周期,共1个周期)治疗。7例接受PCIOD方案患者中,5例(71.43%)达细胞学缓解,2例(28.57%)微小残病灶(MRD)阴性。14例接受VTLP样方案患者中,7例(50.00%)达细胞学缓解,1例(7.14%)MRD阴性。主要不良反应为化疗后骨髓抑制、粒细胞缺乏伴发热、败血症。7例PCIOD方案治疗的患者中3例发生肺部感染,其中1例合并败血症(肺炎克雷伯菌),而14例VTLP样方案治疗的患者中5例发生肺部感染,3例发生败血症,2例发生急性胰腺炎。与传统方案相比,主要不良反应发生率并未增加。所有患者均无治疗相关死亡。结论 ETP-ALL诱导缓解率低,恶性程度高,易早期复发,生存期短。PCIOD方案可以提高ETP-ALL患者的完全缓解率及MRD转阴率,且不增加治疗相关死亡率,但因病例数较少,有待开展多中心、随机对照研究进一步明确。
-
关键词:
- 急性早前T淋巴细胞白血病 /
- 完全缓解 /
- 微小残留病灶
Abstract: Objective There is no standard induction chemotherapy for adult acute lymphoblastic leukemia. Early T-cell precursor acute lymphoblastic leukemia(ETP-ALL) has poor response to standard intensive chemotherapy, high risk of recurrence and poor prognosis. To explore the appropriate induction program and thus improve the remission rate and prolong the survival period.Methods A total of 21 newly diagnosed patients with ETP-ALL who received induction chemotherapy with different regimens from January 2017 to December 2020 were retrospectively analyzed for efficacy and main adverse events. The initial diagnosis of ETP-ALL was based on the results of bone marrow cytology and flow cytometry immunophenotype.Results The 21 patients included 14 males and 7 females, with a median age of 27(16-57) years. Seven patients received PCIOD regimen and 14 patients received VTLP like regimen. Among the 7 patients who received PCIOD regimen, 5 patients(71.43%) achieved cytological response, and 2 patients(28.57%) were minimal residual disease(MRD) negative. In 14 patients who received VTLP-like regimen, 7 patients(50.00%) achieved cytological remission, of which 1 patient(7.14%) was MRD negative. Bone marrow suppression, granulocytopia with fever and septicemia after chemotherapy. Among the 7 patients with PCIOD regimen, 3 patients developed pulmonary infection, including 1 patient with sepsis(klebsiella pneumoniae), while 5 patients with VTLP-like regimen developed pulmonary infection, 3 patients with sepsis, and 2 patients with acute pancreatitis. The incidence of major adverse events was not increased comparing with conventional regimens. There was no treatment-related mortality in any of the patients.Conclusion ETP-ALL has low remission rate of induction therapy and highly malignant character, patients are prone to early recurrence and have short survival. PCIOD regimen can improve the complete response rate and MRD conversion rate of ETP-ALL patients, and does not increase the treatment-related mortality. However, due to the small number of cases, our result needs to be further confirmed by multi-center, randomized controlled studies. -

-
表 1 患者临床特征
例 临床特征 例数 性别 男 14 女 7 年龄/岁 16~34 13 ≥35 8 初诊时外周血WBC/(×109·L-1) >100 2 ≤100 19 初诊时合并中枢神经系统白血病和(或)睾丸白血病 有 1 无 20 纵隔包块 有 3 无 18 细胞遗传学 正常染色体 14 复杂染色体 3 未测 4 初始诱导治疗效果 1个疗程诱导后CR 12 1个疗程诱导后未缓解 9 2个疗程后CR 19 2个疗程后未缓解 1 失访 1 移植 是 14 否 7 表 2 首次诱导及2个疗程后CR率及MRD转阴率
例(%) 组别 例数 诱导后CR MRD阴性 第2个疗程后CR 第2个疗程后MRD阴性 PCIOD 7 5(71.43) 2(28.57) 6(85.71) 5(71.43) VTLP±CTX 14 7(50.00) 1(7.14) 12(85.71) 7(50.00) 表 3 主要不良反应发生率
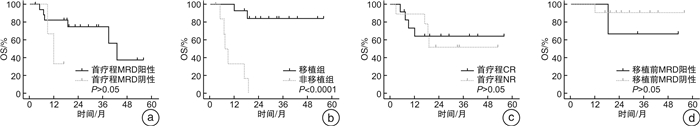
例(%) 主要不良反应 PCIOD方案(7例) VTLP样方案(14例) 败血症 1(14.29) 3(21.43) 粒细胞缺乏伴发热 3(42.86) 9(64.29) 肺部感染 3(42.86) 5(35.71) 急性胰腺炎 0 2(14.29) 肝功能损害 3(42.86) 3(21.43) 尿酸增高 0 3(21.43) 表 4 HSCT可提高ETP-ALL患者长期OS率
诱导化疗 例数(21例) 移植(14例) 未移植(7例) PCIOD方案 7 5例,1例死亡(因严重感染、呼吸衰竭死亡) 2例均死亡 VTLP样方案 14 9例,1例死亡(MRD持续阳性,因复发死亡) 5例(4例死亡,1例失访) -
[1] Coustan-Smith E, Mullighan CG, Onciu M, et al. Early T-cell precursor leukaemia: a subtype of very high-risk acute lymphoblastic leukaemia[J]. Lancet Oncol, 2009, 10(2): 147-156. doi: 10.1016/S1470-2045(08)70314-0
[2] Jain N, Lamb AV, O'Brien S, et al. Early T-cell precursor acute lymphoblastic leukemia/lymphoma(ETP-ALL/LBL)in adolescents and adults: a high-risk subtype[J]. Blood, 2016, 127(15): 1863-1869. doi: 10.1182/blood-2015-08-661702
[3] 黄走方, 王婷玉, 傅明伟, 等. 成人早期前体T急性淋巴细胞白血病的临床特征及预后[J]. 临床血液学杂志, 2018, 31(11): 833-837. doi: 10.13201/j.issn.1004-2806.2018.11.005
[4] Bond J, Graux C, Lhermitte L, et al. Early response-based therapy stratification improves survival in adult early thymic precursor acute lymphoblastic leukemia: a group for research on adult acute lymphoblastic leukemia study[J]. J Clin Oncol, 2017, 35(23): 2683-2691. doi: 10.1200/JCO.2016.71.8585
[5] Ribera JM, Morgades M, Ciudad J, et al. Chemotherapy or allogeneic transplantation in high-risk Philadelphia chromosome-negative adult lymphoblastic leukemia[J]. Blood, 2021, 137(14): 1879-1894. doi: 10.1182/blood.2020007311
[6] Inukai T, Kiyokawa N, Campana D, et al. Clinical significance of early T-cell precursor acute lymphoblastic leukaemia: results of the Tokyo Children's Cancer Study Group Study L99-15[J]. Br J Haematol, 2012, 156(3): 358-365. doi: 10.1111/j.1365-2141.2011.08955.x
[7] Pui CH, Robison LL, Look AT. Acute lymphoblastic leukaemia[J]. Lancet, 2008, 371(9617): 1030-1043. doi: 10.1016/S0140-6736(08)60457-2
[8] Sin CF, Man PM. Early T-Cell Precursor Acute Lymphoblastic Leukemia: Diagnosis, Updates in Molecular Pathogenesis, Management, and Novel Therapies[J]. Front Oncol, 2021, 11: 750789. doi: 10.3389/fonc.2021.750789
[9] Liu Y, Easton J, Shao Y, et al. The genomic landscape of pediatric and young adult T-lineage acute lymphoblastic leukemia[J]. Nat Genet, 2017, 49(8): 1211-1218. doi: 10.1038/ng.3909
[10] 中国抗癌协会血液肿瘤专业委员会, 中华医学会血液学分会白血病淋巴瘤学组. 中国成人急性淋巴细胞白血病诊断与治疗指南(2021年版)[J]. 中华血液学杂志, 2021, 42(9): 705-716. https://www.cnki.com.cn/Article/CJFDTOTAL-LCXZ202203002.htm
[11] 林娜, 刘正华, 夏雪娇, 等. 成人早期前体T急性淋巴细胞白血病(ETP-ALL)与非ETP-ALL的临床特点对比[J]. 现代肿瘤医学, 2019, 27(21): 3875-3877. doi: 10.3969/j.issn.1672-4992.2019.21.033
[12] Koh KN, Im HJ, Kim H, et al. Outcome of Reinduction Chemotherapy with a Modified Dose of Idarubicin for Children with Marrow-Relapsed Acute Lymphoblastic Leukemia: Results of the Childhood Acute Lymphoblastic Leukemia(CALL)-0603 Study[J]. J Korean Med Sci, 2017, 32(4): 642-649. doi: 10.3346/jkms.2017.32.4.642
[13] Weiss MA, Aliff TB, Tallman MS, et al. A single, high dose of idarubicin combined with cytarabine as induction therapy for adult patients with recurrent or refractory acute lymphoblastic leukemia[J]. Cancer, 2002, 95(3): 581-587. doi: 10.1002/cncr.10707
[14] Zhou L, Liu X, Liu H, et al. A comparative study of idarubicin 12 mg/m2and 8 mg/m2combined with cytarabine as the first induction regimen for adult acute myeloid leukemia patients[J]. Onco Targets Ther, 2016, 9: 985-991.
[15] Schwartz CL, Thompson EB, Gelber RD, et al. Improved response with higher corticosteroid dose in children with acute lymphoblastic leukemia[J]. J Clin Oncol, 2001, 19(4): 1040-1046. doi: 10.1200/JCO.2001.19.4.1040
[16] Zhang Y, Qian JJ, Zhou YL, et al. Comparison of Early T-Cell Precursor and Non-ETP Subtypes Among 122 Chinese Adults With Acute Lymphoblastic Leukemia[J]. Front Oncol, 2020, 10: 1423. doi: 10.3389/fonc.2020.01423
[17] Neumann M, Heesch S, Gökbuget N, et al. Clinical and molecular characterization of early T-cell precursor leukemia: a high-risk subgroup in adult T-ALL with a high frequency of FLT3 mutations[J]. Blood Cancer J, 2012, 2(1): e55. doi: 10.1038/bcj.2011.49
[18] Maude SL, Dolai S, Delgado-Martin C, et al. Efficacy of JAK/STAT pathway inhibition in murine xenograft models of early T-cell precursor(ETP)acute lymphoblastic leukemia[J]. Blood, 2015, 125(11): 1759-1767. doi: 10.1182/blood-2014-06-580480
[19] Bernt KM, Neff T. The role of polycomb repressive complex 2 in early T-cell precursor acute lymphoblastic leukemia[J]. Mol Cell Oncol, 2018, 5(5): e1166309. doi: 10.1080/23723556.2016.1166309
[20] Bernt KM, Hunger SP, Neff T. The Functional Role of PRC2 in Early T-cell Precursor Acute Lymphoblastic Leukemia(ETP-ALL)-Mechanisms and Opportunities[J]. Front Pediatr, 2016, 4: 49.
[21] Chonghaile TN, Roderick JE, Glenfield C, et al. Maturation stage of T-cell acute lymphoblastic leukemia determines BCL-2 versus BCL-XL dependence and sensitivity to ABT-199[J]. Cancer Discov, 2014, 4(9): 1074-1087. doi: 10.1158/2159-8290.CD-14-0353
[22] Peirs S, Matthijssens F, Goossens S, et al. ABT-199 mediated inhibition of BCL-2 as a novel therapeutic strategy in T-cell acute lymphoblastic leukemia[J]. Blood, 2014, 124(25): 3738-3747. doi: 10.1182/blood-2014-05-574566
[23] Hoelzer D, Thiel E, Arnold R, et al. Sucessful subtype oriented treatment strategies in adult T-ALL: results of 744 patients treated in three consecutive GMALL studies[J]. Blood, 2009, 144: 324-324. doi: 10.3760/cma.j.issn.1673-419X.2009.04.009
[24] Marks DI, Paietta EM, Moorman AV, et al. T-cell acute lymphoblastic leukemia in adults: clinical features, immunophenotype, cytogenetics, and outcome from the large randomized prospective trial(UKALL Ⅻ/ECOG 2993)[J]. Blood, 2009, 114(25): 5136-5145. doi: 10.1182/blood-2009-08-231217
[25] 赵艳丽, 陆佩华. CAR-T细胞治疗在急性B细胞淋巴细胞白血病异基因造血干细胞移植中的应用[J]. 临床血液学杂志, 2021, 34(9): 603-607. https://www.cnki.com.cn/Article/CJFDTOTAL-LCXZ202109001.htm
[26] 张宜婧, 沈利, 向春丽, 等. 成人急性淋巴细胞白血病免疫治疗进展[J]. 临床血液学杂志, 2022, 35(1): 82-86. http://lcxz.cbpt.cnki.net/WKC/WebPublication/paperDigest.aspx?paperID=b2a29548-daa2-448a-9f7a-dc60fa0c2f6e
-




 下载:
下载: