-
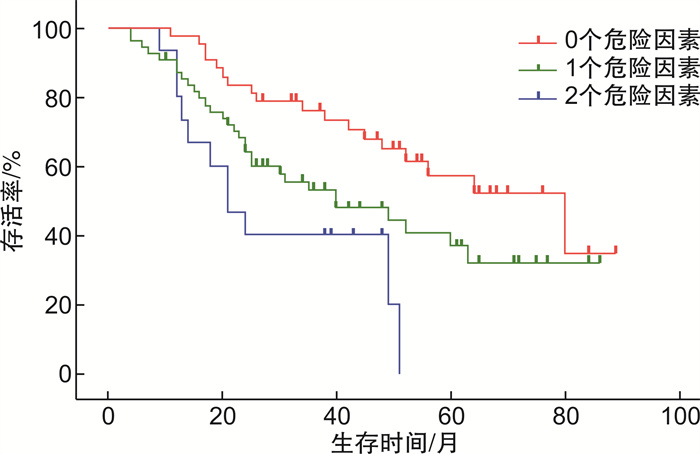
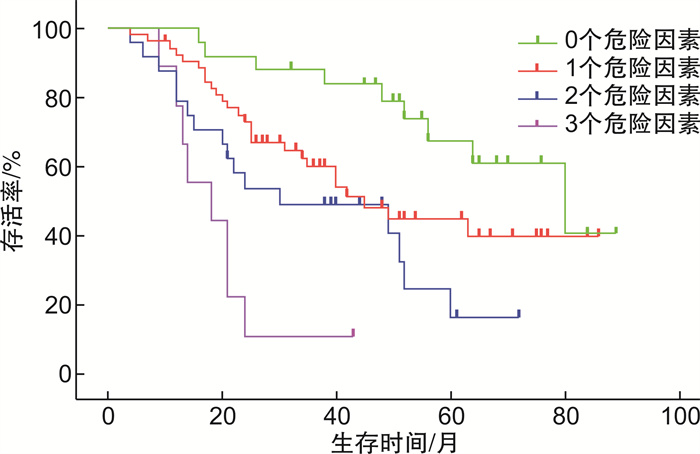
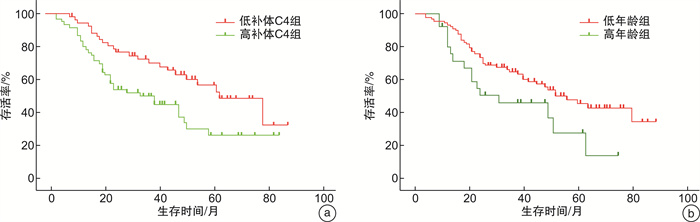
摘要: 目的 探讨补体C4对新诊断多发性骨髓瘤(MM)患者生存预后的价值。方法 回顾性分析2015年1月—2020年6月在江苏省苏北人民医院确诊的111例MM患者的临床资料,通过Cox回归分析对其生存预后进行单因素及多因素分析,通过logistic回归分析对2年生存期进行单因素及多因素分析。结果 111例MM患者中男59例,女52例,中位年龄64(34~86)岁,总体中位生存期为51个月。通过Cox回归分析进行生存预后分析,结果显示补体C4水平及年龄为影响预后的独立因素(P < 0.05)。通过logistic回归分析对2年生存期进行分析,结果显示补体C4≥0.2 mg/mL、年龄≥70岁及白细胞计数≤4.4×109/L为独立预后因素,同时存在3个危险因素的患者2年内死亡的风险是存在0~2个危险因素患者的8倍(P < 0.05)。结论 高补体C4水平的患者生存预后差,补体C4水平可以作为MM患者生存预后的潜在生物学标记物。Abstract: Objective To explore the predictive value of completement C4 in newly diagnosed multiple myeloma(MM).Methods The clinical data of 111 newly diagnosed MM were retrospectively analyzed. The univariate and multivariate analysis for overall survival(OS) were made through Cox regression analysis and univariate and multivariate analysis for two-year survival were made through logistic regression analysis.Results Among the 111 patients, there were 59 males and 52 females, with a medium age of 64(34-86) years and the medium OS was 51 months. It revealed that high C4 level and advanced age were the independent factors of OS through Cox regression analysis(P < 0.05). C4≥0.2 mg/mL, age≥70 years and WBC≤4.4×109/L were independent factors for two-year survival through logistic regression analysis(P < 0.05). It indicated that patients with 3 risk factors had 8 folds of mortality risk in two years than patients with 0-2 risk factors(P < 0.05).Conclusion Patients with high level C4 have poor OS and C4 can be the potential biomarker for OS of newly diagnosed MM.
-
Key words:
- multiple myeloma /
- completement C4 /
- survival analysis
-

-
表 1 111例患者的临床资料及影响OS的单因素及多因素分析
临床特征 总例数(n=111) HR 95%CI P 单因素分析 性别/例(%) 0.810 0.484~1.354 0.421 男 59(53.15) 女 52(46.85) 年龄/岁 62.66±9.68 1.045 1.016~1.075 0.002 血红蛋白/(g·L-1) 86.11±25.42 1.001 0.991~1.011 0.800 白细胞计数/(×109·L-1) 5.04±2.08 0.814 0.693~0.956 0.007 中性粒细胞/(×109·L-1) 3.70±4.75 1.017 0.960~1.078 0.560 淋巴细胞/(×109·L-1) 1.34±0.66 0.519 0.304~0.886 0.016 血小板计数/(×109·L-1) 160.23±74.92 0.997 0.993~1.001 0.139 白蛋白/(g·L-1) 36.01±7.00 1.022 0.985~1.061 0.249 球蛋白/(g·L-1) 53.23±30.18 0.991 0.981~1.000 0.047 A/G 1.06±0.84 1.241 0.937~1.643 0.132 LDH/(U·L-1) 202.54±84.30 1.003 1.000~1.006 0.081 血清钙/(mmol·L-1) 2.38±0.44 1.038 0.576~1.872 0.901 血清校正钙/(mmol·L-1) 2.45±0.44 0.999 0.555~1.798 0.997 血肌酐/(μmol·L-1) 151.37±172.39 1.001 1.000~1.002 0.190 血清β2-MG/(μg·mL-1) 4.72±4.13 1.040 0.979~1.105 0.204 尿β2-MG/(μg·mL-1) 2.21±3.54 1.015 0.934~1.013 0.724 补体C3/(mg·mL-1) 0.94±0.27 1.152 0.435~3.052 0.776 补体C4/(mg·mL-1) 0.23±0.17 4.396 1.077~17.941 0.039 M蛋白/(g·L-1) 30.32±20.64 0.995 0.981~1.010 0.547 骨髓浆细胞比例/% 33.75±20.91 1.006 0.996~1.017 0.248 ISS分期/例(%) Ⅰ 39(35.14) 0.307 Ⅱ 44(39.64) 0.740 0.403~1.361 0.333 Ⅲ 28(25.22) 1.243 0.657~2.351 0.504 一线治疗方案/例(%) 含硼替佐米 95(85.59) 1.048 0.515~2.135 0.897 不含硼替佐米 16(14.41) 多因素分析 年龄 1.044 1.013~1.076 0.005 补体C4/(mg·mL-1) 5.994 1.309~27.442 0.021 表 2 影响患者2年生存期的单因素分析
参数 2年生存组(n=77) 2年死亡组(n=34) HR(95%CI) P 性别/例 0.603(0.265~1.374) 0.229 男 38 21 女 39 13 年龄/岁 61.23±9.91 65.88±8.40 1.057(1.008~0.018) 0.022 血红蛋白/(g·L-1) 87.25±24.03 83.53±28.53 0.994(0.978~1.010) 0.477 白细胞计数/(×109·L-1) 5.14±2.16 4.12±1.52 0.643(0.480~0.862) 0.003 中性粒细胞/(×109·L-1) 3.54±1.74 2.72±1.30 1.012(0.943~1.106) 0.608 淋巴细胞/(×109·L-1) 1.44±0.68 1.13±0.56 0.394(0.171~0.910) 0.029 血小板计数/(×109·L-1) 170.60±75.74 136.74±68.40 0.993(0.987~0.999) 0.031 白蛋白/(g·L-1) 35.96±6.69 36.14±7.78 1.004(0.947~1.064) 0.889 球蛋白/(g·L-1) 55.01±32.25 49.19±24.86 0.993(0.980~1.007) 0.348 A/G 1.06±0.87 1.06±0.78 0.996(0.615~1.613) 0.985 LDH/(U·L-1) 195.32±76.86 218.68±98.28 1.003(0.998~1.008) 0.185 血清钙/(mmol·L-1) 2.36±0.39 2.43±0.54 1.410(0.576~3.450) 0.451 血清校正钙/(mmol·L-1) 2.43±0.40 2.51±0.54 1.520(0.641~3.763) 0.365 血肌酐/(μmol·L-1) 139.64±142.74 178.73±227.46 1.001(0.999~1.003) 0.285 血清β2-MG/(μg·mL-1) 4.63±3.94 4.92±4.59 1.017(0.922~1.122) 0.740 尿β2-MG/(μg·mL-1) 1.85±2.63 3.01±5.01 1.090(0.957~1.241) 0.197 补体C3/(mg·mL-1) 0.94±0.28 0.93±0.27 0.772(0.174~3.421) 0.734 补体C4/(mg·mL-1) 0.21±0.15 0.28±0.20 11.775(1.088~127.462) 0.042 M蛋白/(g·L-1) 36.50±21.40 39.40±18.85 1.007(0.984~1.031) 0.558 骨髓浆细胞比例/% 31.69±21.11 38.50±19.95 1.015(0.996~1.036) 0.127 ISS/例(%) Ⅰ 27(24.32) 12(10.81) 0.958 Ⅱ 30(27.03) 14(12.61) 1.050(0.414~2.661) 0.918 Ⅲ 20(18.02) 8(7.21) 0.900(0.310~2.611) 0.846 表 3 影响患者2年生存期的多因素分析

-
[1] 刘志月, 刘梅, 黄鹤, 等. 实时三维超声心动图及三维斑点追踪技术评估左心室射血分数保留的多发性骨髓瘤患者左心房功能的价值[J]. 临床心血管病杂志, 2021, 37(10): 936-942. https://www.cnki.com.cn/Article/CJFDTOTAL-LCXB202110013.htm
[2] Atkin C, Iqbal G, Planche T, et al. Diagnostic pathways in multiple myeloma and their relationship to end organ damage: an analysis from the Tackling Early Morbidity and Mortality in Myeloma(TEAMM)trial[J]. Br J Haematol, 2021, 192(6): 997-1005. doi: 10.1111/bjh.17044
[3] Palumbo A, Avet-Loiseau H, Oliva S, et al. Revised International Staging System for Multiple Myeloma: A Report From International Myeloma Working Group[J]. J Clin Oncol, 2015, 33(26): 2863-2869. doi: 10.1200/JCO.2015.61.2267
[4] Dowling P, Hayes C, Ting KR, et al. Identification of proteins found to be significantly altered when comparing the serum proteome from Multiple Myeloma patients with varying degrees of bone disease[J]. BMC Genomics, 2014, 15: 904. doi: 10.1186/1471-2164-15-904
[5] Zhang L, Ling X, Li F, et al. Complement 4 Aids in the Prediction of Newly Diagnosed Multiple Myeloma Outcome in Patients[J]. Clin Med Insights Oncol, 2022, 16: 11795549221079171.
[6] 黄晓军, 路瑾, 侯健, 等. 中国多发性骨髓瘤诊治指南(2015年修订)[J]. 中华内科杂志, 2015, 54(12): 1066-1070. https://www.cnki.com.cn/Article/CJFDTOTAL-JLYB201610002.htm
[7] Walport MJ. Complement. First of two parts[J]. N Engl J Med, 2001, 344(14): 1058-1066. doi: 10.1056/NEJM200104053441406
[8] Wang Y, Zhou S, Wang D, et al. Complement C4-A and Plasminogen as Potential Biomarkers for Prediction of Papillary Thyroid Carcinoma[J]. Front Endocrinol(Lausanne), 2021, 12: 737638. doi: 10.3389/fendo.2021.737638
[9] Roumenina LT, Daugan MV, Noé R, et al. Tumor Cells Hijack Macrophage-Produced Complement C1q to Promote Tumor Growth[J]. Cancer Immunol Res, 2019, 7(7): 1091-1105. doi: 10.1158/2326-6066.CIR-18-0891
[10] Daugan MV, Revel M, Russick J, et al. Complement C1s and C4d as Prognostic Biomarkers in Renal Cancer: Emergence of Noncanonical Functions of C1s[J]. Cancer Immunol Res, 2021, 9(8): 891-908. doi: 10.1158/2326-6066.CIR-20-0532
[11] Ajona D, Pajares MJ, Corrales L, et al. Investigation of complement activation product c4d as a diagnostic and prognostic biomarker for lung cancer[J]. J Natl Cancer Inst, 2013, 105(18): 1385-1393. doi: 10.1093/jnci/djt205
[12] Ajona D, Okrój M, Pajares MJ, et al. Complement C4d-specific antibodies for the diagnosis of lung cancer[J]. Oncotarget, 2018, 9(5): 6346-6355. doi: 10.18632/oncotarget.23690
[13] Klikovits T, Stockhammer P, Laszlo V, et al. Circulating complement component4d(C4d)correlates with tumor volume, chemotherapeutic response and survival in patients with malignant pleural mesothelioma[J]. Sci Rep, 2017, 7(1): 16456. doi: 10.1038/s41598-017-16551-7
[14] Reis ES, Mastellos DC, Ricklin D, et al. Complement in cancer: untangling an intricate relationship[J]. Nat Rev Immunol, 2018, 18(1): 5-18. doi: 10.1038/nri.2017.97
[15] Cho MS, Vasquez HG, Rupaimoole R, et al. Autocrine effects of tumor-derived complement[J]. Cell Rep, 2014, 6(6): 1085-1095. doi: 10.1016/j.celrep.2014.02.014
[16] Minnie SA, Hill GR. Immunotherapy of multiple myeloma[J]. J Clin Invest, 2020, 130(4): 1565-1575. doi: 10.1172/JCI129205
[17] 邝丽芬, 房佰俊, 陈文明, 等. 含塞利尼索方案治疗复发难治多发性骨髓瘤的疗效与安全性分析: 一项中国多中心真实世界研究[J]. 临床血液学杂志, 2022, 35(9): 626-632. https://lcxy.whuhzzs.com/article/doi/10.13201/j.issn.1004-2806.2022.09.005
[18] Rodriguez-Otero P, Paiva B, San-Miguel JF. Roadmap to cure multiple myeloma[J]. Cancer Treat Rev, 2021, 100: 102284. doi: 10.1016/j.ctrv.2021.102284
[19] Moreau P, Cavo M, Sonneveld P, et al. Combination of international scoring system 3, high lactate dehydrogenase, and t(4;14) and/or del(17p)identifies patients with multiple myeloma(MM)treated with front-line autologous stem-cell transplantation at high risk of early MM progression-related death[J]. J Clin Oncol, 2014, 32(20): 2173-2180. doi: 10.1200/JCO.2013.53.0329
[20] Rodríguez-Otero P, Mateos MV, Martínez-López J, et al. Early myeloma-related death in elderly patients: development of a clinical prognostic score and evaluation of response sustainability role[J]. Leukemia, 2018, 32(11): 2427-2434. doi: 10.1038/s41375-018-0072-6
[21] Luo S, Wang M, Wang H, et al. How Does Complement Affect Hematological Malignancies: From Basic Mechanisms to Clinical Application[J]. Front Immunol, 2020, 11: 593610. doi: 10.3389/fimmu.2020.593610
[22] van de Donk NW, Janmaat ML, Mutis T, et al. Monoclonal antibodies targeting CD38 in hematological malignancies and beyond[J]. Immunol Rev, 2016, 270(1): 95-112. doi: 10.1111/imr.12389
[23] Abramson HN. Monoclonal Antibodies for the Treatment of Multiple Myeloma: An Update[J]. Int J Mol Sci, 2018, 19(12): 3924.
[24] Nishida H, Yamada T. Monoclonal Antibody Therapies in Multiple Myeloma: A Challenge to Develop Novel Targets[J]. J Oncol, 2019, 2019: 6084012.
[25] Chang-Han L, Gabrielle R, Wupeng Y, et al. IgG Fc domains that bind C1q but not effector Fcγ receptors delineate the importance of complement-mediated effector functions[J]. Nat Immunol, 2017, 18(8): 889-898.
-

| 引用本文: | 冷晴, 刘伟, 管俊. 补体C4在新诊断多发性骨髓瘤患者中的预后价值[J]. 临床血液学杂志, 2023, 36(3): 175-180. doi: 10.13201/j.issn.1004-2806.2023.03.007 |
| Citation: | LENG Qing, LIU Wei, GUAN Jun. The value of completement C4 in newly diagnosed multiple myeloma[J]. J Clin Hematol, 2023, 36(3): 175-180. doi: 10.13201/j.issn.1004-2806.2023.03.007 |
- Figure 1.
- Figure 2.
- Figure 3.



 下载:
下载: