The pathogenesis of a female hemophilia A patient with heterozygous variant and literature review
-
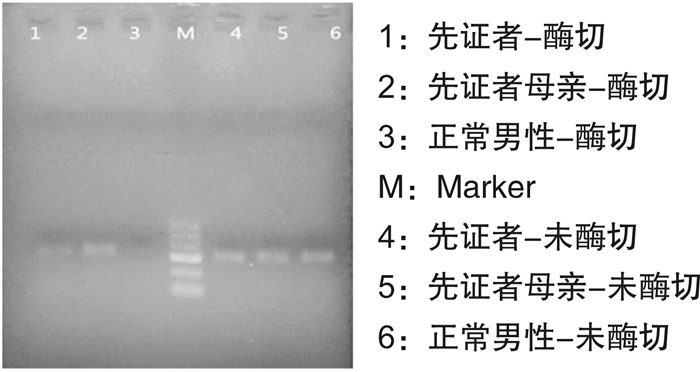
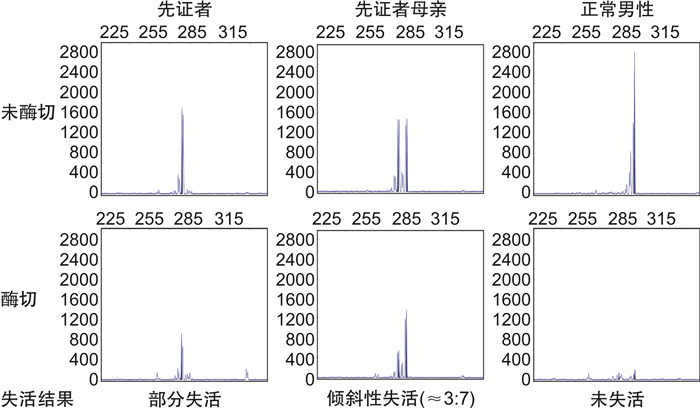
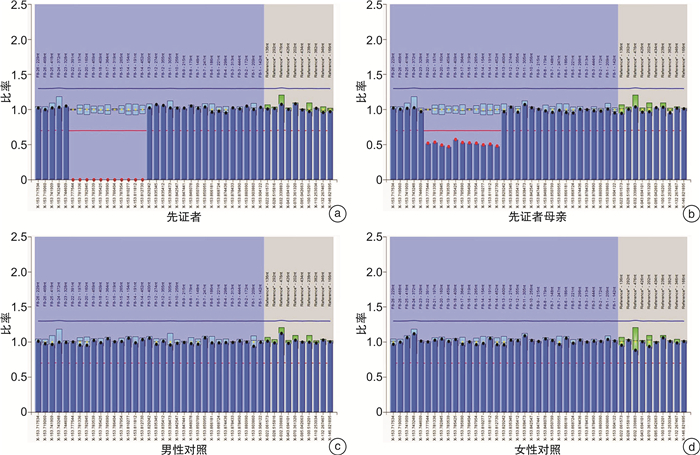
摘要: 通过对血友病A(hemophilia A,HA)患者家系进行临床特征分析和分子学检测,探讨女性FⅧ基因杂合变异致轻型HA的发病机制,并通过检索国际FⅧ基因变异数据库进行相关文献复习。结果显示先证者FⅧ:C 0.3%,为重型HA患者,伴有抑制物产生,基因变异检测发现先证者存在大片段缺失变异(c.delexons14_22),此变异为已知的致病性变异,目前国内外报道了6例患者,均为重型HA,其中5例报道了抑制物产生,1例患者未对抑制物进行描述。先证者母亲为大片段缺失变异的杂合变异,FⅧ:C 13%,为轻型HA,未见抑制物形成;X染色体失活检测发现该母亲的正常X染色体比携带FⅧ基因变异的X染色体表达活性低。大片段缺失变异(c.delexons14_22)可引起重型HA,且与抑制物产生高度相关。女性杂合变异致轻型HA的机制可解释为X染色体表达存在非随机不平衡失活。Abstract: To explore the pathogenetic mechanism for a female patient affected with mild hemophilia A caused by heterozygous variants in FⅧ genes, it analyzed the clinical characteristics and molecular characteristics of the patients' families. Relevant literature was reviewed by searching the International FⅧ Variant Database. The proband, a 22-year-old boy was diagnosed with severe HA at 1-year-old, Sanger sequencing failed to identify molecular defects, and MLPA revealed a large duplication (c. delexons14_22). This variant has been observed in 6 HA patients, 5 of whom had severe phenotype and had a history of inhibitors. His mother was a carrier of large duplication (c. delexons14_22), with a lower concentration of FⅧ(FⅧ: C 13%). X chromosome inactivation test found that the normal X chromosome of the mother had lower expression activity than the X chromosome carrying the FⅧ gene variant. Large duplications (c. delexons14_22) can cause severe HA and is highly correlated with inhibitor production. The mechanism of mild HA induced by heterozygous variant can be explained by the non-random imbalance inactivation of the X chromosome.
-
Key words:
- femininity /
- hemophilia A /
- FⅧ gene /
- large deletion /
- X chromosome inactivation
-

-
[1] Mahlangu J, Oldenburg J, Paz-Priel I, et al. Emicizumab prophylaxis in patients who have hemophilia A without inhibitors[J]. N Engl J Med, 2018, 379(9): 811-822. doi: 10.1056/NEJMoa1803550
[2] Hirayama AB, Silva AKCD, Rocha JS, et al. Prevalence of symptoms in hemophilia carriers in comparison with the general population: a systematic review[J]. Hematol Transfus Cell Ther, 2019, 41(4): 349-355. doi: 10.1016/j.htct.2019.02.006
[3] Al-Huniti A, Sharathkumar A, Krantz M, et al. Discrepant hemophilia A: an underdiagnosed disease entity[J]. Am J Clin Pathol, 2020, 154(1): 78-87. doi: 10.1093/ajcp/aqaa024
[4] Seaman CD, Xavier F, Ragni MV. Hemophilia A(factor Ⅷ deficiency)[J]. Hematol Oncol Clin North Am, 2021, 35(6): 1117-1129. doi: 10.1016/j.hoc.2021.07.006
[5] Franchini M, Mannucci PM. The history of hemophilia[J]. Semin Thromb Hemost, 2014, 40(5): 571-576. doi: 10.1055/s-0034-1381232
[6] Mannucci PM. Hemophilia therapy: the future has begun[J]. Haematologica, 2020, 105(3): 545-553. doi: 10.3324/haematol.2019.232132
[7] 张磊, 代新岳. 血友病基因治疗临床研究进展[J]. 临床血液学杂志, 2022, 35(7): 464-468. doi: 10.13201/j.issn.1004-2806.2022.07.003 https://lcxy.whuhzzs.com/article/doi/10.13201/j.issn.1004-2806.2022.07.003
[8] Shinozawa K, Amano K, Hagiwara T, et al. Genetic analysis of carrier status in female members of Japanese hemophilia families[J]. J Thromb Haemost, 2021, 19(6): 1493-1505. doi: 10.1111/jth.15301
[9] Paroskie A, Gailani D, DeBaun MR, et al. A cross-sectional study of bleeding phenotype in haemophilia A carriers[J]. Br J Haematol, 2015, 170(2): 223-228. doi: 10.1111/bjh.13423
[10] Garagiola I, Mortarino M, Siboni SM, et al. X Chromosome inactivation: a modifier of factor Ⅷ and Ⅸ plasma levels and bleeding phenotype in Haemophilia carriers[J]. Eur J Hum Genet, 2021, 29(2): 241-249. doi: 10.1038/s41431-020-00742-4
[11] 中华医学会血液学分会血栓与止血学组, 中国血友病协作组. 凝血因子Ⅷ/Ⅸ抑制物诊断与治疗中国指南(2018年版)[J]. 中华血液学杂志, 2018, 39(10): 793-799. doi: 10.3760/cma.j.issn.0253-2727.2018.10.001
[12] Spena S, Garagiola I, Cannavò A, et al. Prediction of factor Ⅷ inhibitor development in the SIPPET cohort by mutational analysis and factor Ⅷ antigen measurement[J]. J Thromb Haemost, 2018, 16(4): 778-790. doi: 10.1111/jth.13961
[13] Cygan PH, Kouides PA. Regulation and importance of factor ⅧI levels in hemophilia A carriers[J]. Curr Opin Hematol, 2021, 28(5): 315-322. doi: 10.1097/MOH.0000000000000667
[14] Plug I, Mauser-Bunschoten EP, Bröcker-Vriends AHJT, et al. Bleeding in carriers of hemophilia[J]. Blood, 2006, 108(1): 52-56. doi: 10.1182/blood-2005-09-3879
[15] Miller CH, Bean CJ. Genetic causes of haemophilia in women and girls[J]. Haemophilia, 2021, 27(2): e164-e179.
[16] d'Oiron R, O'Brien S, James AH. Women and girls with haemophilia: lessons learned[J]. Haemophilia, 2021, 27(Suppl 3): 75-81.
[17] Loda A, Collombet S, Heard E. Gene regulation in time and space during X-chromosome inactivation[J]. Nat Rev Mol Cell Biol, 2022, 23(4): 231-249.
[18] Furlan G, Galupa R. Mechanisms of choice in X-chromosome inactivation[J]. Cells, 2022, 11(3): 535. doi: 10.3390/cells11030535
[19] Wang D, Tang L, Wu YF, et al. Abnormal X chromosome inactivation and tumor development[J]. Cell Mol Life Sci, 2020, 77(15): 2949-2958. doi: 10.1007/s00018-020-03469-z
[20] Pereira G, Dória S. X-chromosome inactivation: implications in human disease[J]. J Genet, 2021, 100: 63. doi: 10.1007/s12041-021-01314-1
[21] Dossin F, Heard E. The molecular and nuclear dynamics of X-chromosome inactivation[J]. Cold Spring Harb Perspect Biol, 2022, 14(4): a040196.
[22] Shvetsova E, Sofronova A, Monajemi R, et al. Skewed X-inactivation is common in the general female population[J]. Eur J Hum Genet, 2019, 27(3): 455-465. doi: 10.1038/s41431-018-0291-3
[23] Posynick BJ, Brown CJ. Escape from X-chromosome inactivation: an evolutionary perspective[J]. Front Cell Dev Biol, 2019, 7: 241. doi: 10.3389/fcell.2019.00241
[24] Vianna EQ, Piergiorge RM, Gonçalves AP, et al. Understanding the landscape of X-linked variants causing intellectual disability in females through extreme X chromosome inactivation skewing[J]. Mol Neurobiol, 2020, 57(9): 3671-3684. doi: 10.1007/s12035-020-01981-8
[25] Dardik R, Avishai E, Lalezari S, et al. Molecular mechanisms of skewed X-chromosome inactivation in female hemophilia patients-lessons from wide genome analyses[J]. Int J Mol Sci, 2021, 22(16): 9074. doi: 10.3390/ijms22169074
-




 下载:
下载: