Effects of different storage conditions and different sample types on concentration analysis of sCD40L in blood samples
-
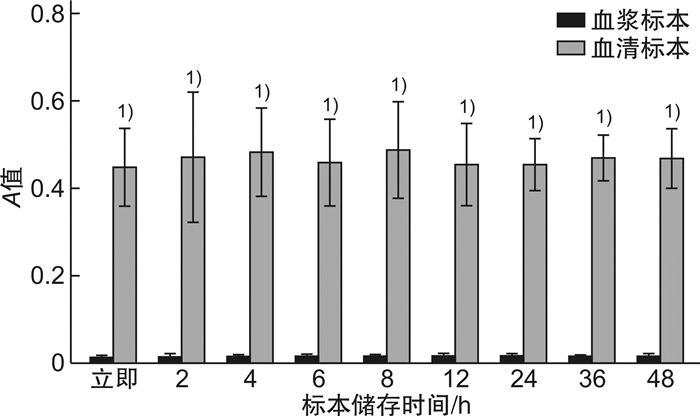
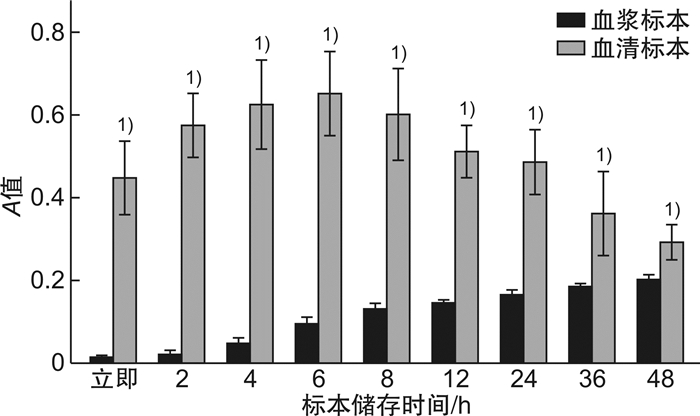
摘要: 目的 探究不同储存条件及不同样本类型对血样中可溶性白细胞分化抗原CD40配体(sCD40L)浓度的影响,为临床更精确检测sCD40L提供保障。方法 随机选取正常人的血浆和血清标本,分别在4℃和室温下储存0、2、4、8、16、24、36、48 h,利用ELISA方法检测其中sCD40L的表达水平。结果 室温及4℃条件下,血清中sCD40L浓度显著高于其血浆中sCD40L浓度(P<0.05);4℃条件下,同一检测者血浆和血清中sCD40L浓度随放置时间变化差异无统计学意义(P>0.05);室温条件下,血浆内sCD40L浓度随放置时间延长而升高,血清中sCD40L浓度随时间变化先升高再降低。结论 sCD40L作为人体一种重要的炎性因子,在其精确检测的过程中,要确定待检标本是血清或血浆及不同的储存时间。Abstract: Objective To explore the effects of different storage conditions and different sample types on the concentration of soluble leukocyte differentiation antigen CD40Ligand(sCD40L) in blood samples, so as to provide guarantee for more accurate clinical detection of sCD40L.Methods Normal plasma and serum samples were randomly selected and stored at 4℃ and room temperature for 0, 2, 4, 8, 16, 24, 36, 48 h, respectively. The expression level of sCD40L was detected by ELISA.Results At room temperature and 4℃, serum sCD40L concentration was significantly higher than plasma sCD40L concentration(P < 0.05). At 4℃, sCD40L concentration in plasma and serum of the same test subjects had no significant difference with the time of placement(P>0.05). At room temperature, plasma sCD40L concentration increased with time, while serum sCD40L concentration first increased and then decreased with time.Conclusion sCD40L is an important inflammatory factor in human body. In the process of its accurate detection, it is necessary to determine whether the specimen to be tested is serum or plasma and the different storage time.
-
Key words:
- sCD40L /
- platelets /
- ELISA /
- concentration analysis
-

-
表 1 不同温度储存条件下,血浆及血清中sCD40L(测量A值)随时间变化差异
X±S 不同时间 室温保存血浆标本 室温保存血清标本 4℃保存血浆标本 4℃保存血清标本 对照组(0 h) 0.015±0.003 0.448±0.090 0.015±0.003 0.448±0.090 2 h 0.022±0.008 0.574±0.076 0.017±0.004 0.475±0.144 4 h 0.047±0.0131) 0.625±0.107 0.017±0.002 0.484±0.101 6 h 0.097±0.0151) 0.652±0.102 0.017±0.003 0.461±0.098 8 h 0.133±0.0131) 0.603±0.111 0.016±0.001 0.489±0.109 12 h 0.148±0.0061) 0.512±0.062 0.018±0.004 0.454±0.095 24 h 0.166±0.0101) 0.486±0.079 0.018±0.003 0.457±0.057 36 h 0.189±0.0031) 0.362±0.101 0.018±0.001 0.473±0.050 48 h 0.202±0.0121) 0.292±0.041 0.018±0.003 0.472±0.067 与对照组比较,1)P<0.05。 -
[1] Alaaeddine N, Hassan GS, Yacoub D, et al. CD154: an immunoinflammatory mediator in systemic lupus erythematosus and rheumatoid arthritis[J]. Clin Dev Immunol, 2012, 2012: 490148.
[2] Patoulias D, Stavropoulos K, Imprialos K, et al. Inflammatory markers in cardiovascular disease; lessons learned and future perspectives[J]. Curr Vasc Pharmacol, 2021, 19(3): 323-342.
[3] Henn V, Steinbach S, Büchner K, et al. The inflammatory action of CD40Ligand(CD154) expressed on activated human platelets is temporally limited by coexpressed CD40[J]. Blood, 2001, 98(4): 1047-1054. doi: 10.1182/blood.V98.4.1047
[4] Jensen J, Krakauer M, Sellebjerg F. Increased T cell expression of CD154(CD40-ligand)in multiple sclerosis[J]. Eur J Neurol, 2001, 8(4): 321-328. doi: 10.1046/j.1468-1331.2001.00232.x
[5] Antoniades C, Bakogiannis C, Tousoulis D, et al. The CD40/CD40Ligand system: linking inflammation with atherothrombosis[J]. J Am Coll Cardiol, 2009, 54(8): 669-677. doi: 10.1016/j.jacc.2009.03.076
[6] Aloui C, Prigent A, Sut C, et al. The signaling role of CD40Ligand in platelet biology and in platelet component transfusion[J]. Int J Mol Sci, 2014, 15(12): 22342-22364. doi: 10.3390/ijms151222342
[7] Tascanov MB. May cystatin-c be associated with post-myocardial infarction complications?[J]. Ann Clin Anal Med, 2019, 10(1): 358-368.
[8] Hassan GS, Merhi Y, Mourad W. CD40Ligand: a neo-inflammatory molecule in vascular diseases[J]. Immunobiology, 2012, 217(5): 521-532. doi: 10.1016/j.imbio.2011.03.010
[9] Seigner J, Basilio J, Resch U, et al. CD40L and TNF both activate the classical NF-κB pathway, which is not required for the CD40L induced alternative pathway in endothelial cells[J]. Biochem Biophys Res Commun, 2018, 495(1): 1389-1394. doi: 10.1016/j.bbrc.2017.11.160
[10] 原达, 朱国斌. 血小板P选择素及CD40/CD40L在动脉粥样硬化中的研究进展[J]. 心血管病学进展, 2021, 42(10): 928-932. https://www.cnki.com.cn/Article/CJFDTOTAL-XXGB202110020.htm
[11] Scheinowitz M, Pakala R, Ben-Dor I, et al. Platelet reactivity in diabetic patients subjected to acute exercise stress test[J]. Cardiovasc Revasc Med, 2011, 12(1): 20-24. doi: 10.1016/j.carrev.2010.01.005
[12] 潘宗岱, 薛静, 孙士鹏, 等. 血小板及其衍生物的保存方案和应用进展[J]. 临床血液学杂志, 2022, 35(12): 900-904. https://lcxy.whuhzzs.com/article/doi/10.13201/j.issn.1004-2806.2022.12.015
[13] 姚勇, 张欢欢, 孙振超, 等. 单采血小板保存期内活化状态及相关功能和代谢指标检测及分析[J]. 中国输血杂志, 2021, 34(2): 120-123. https://www.cnki.com.cn/Article/CJFDTOTAL-BLOO202102007.htm
[14] 李文娟, 何芮, 尹湧华, 等. 4℃冷藏保存血小板的储存损伤与应用研究进展[J]. 中国输血杂志, 2021, 34(8): 926-930.
[15] Wu XW, Darlington DN, Montgomery RK, et al. Platelets derived from fresh and cold-stored whole blood participate in clot formation in rats with acute traumatic coagulopathy[J]. Br J Haematol, 2017, 179(5): 802-810. doi: 10.1111/bjh.14999
[16] 罗娜维. 机采血小板保存期内血小板损伤与输注效果的相关性[J]. 临床医学, 2021, 41(6): 47-49.
[17] Stubbs JR, Tran SA, Emery RL, et al. Cold platelets for trauma-associated bleeding: regulatory approval, accreditation approval, and practice implementation-just the "tip of the iceberg"[J]. Transfusion, 2017, 57(12): 2836-2844.
[18] Montgomery RK, Reddoch KM, Evani SJ, et al. Enhanced shear-induced platelet aggregation due to low-temperature storage[J]. Transfusion, 2013, 53(7): 1520-1530.
[19] 李玮, 谢如锋, 任亚娜, 等. 库存血小板血浆中sCD40L含量变化及其对PMNs呼吸爆发的作用[J]. 中国输血杂志, 2013, 26(5): 430-434. https://www.cnki.com.cn/Article/CJFDTOTAL-BLOO201305007.htm
[20] Damien P, Cognasse F, Eyraud MA, et al. LPS stimulation of purified human platelets is partly dependent on plasma soluble CD14 to secrete their main secreted product, soluble-CD40-Ligand[J]. BMC Immunol, 2015, 16(1): 3.
[21] 周明, 陈洋, 王敏, 等. 机采血小板储存损伤对临床疗效的影响[J]. 临床输血与检验, 2019, 21(2): 5.
[22] 常远, 高伟, 田冰, 等. 对血浆中sCD40L的诊断分析[J]. 健康大视野, 2021, (8): 210.
-




 下载:
下载: