Comparative analysis of WBCDouble and WBCPeak in predicting differentiation syndrome during the single agent arsenic trioxide treatment of initial high-risk acute promyelocytic leukemia
-
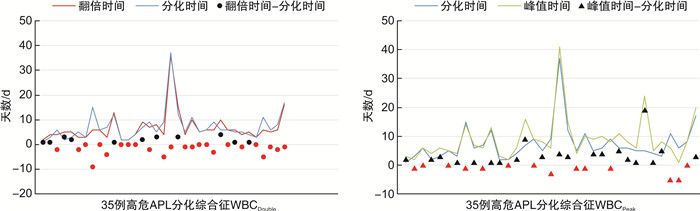
摘要: 目的 筛选预测初发高危急性早幼粒细胞白血病(acute promyelocytic leukemia,APL)发生分化综合征的因素,比较翻倍白细胞(WBCDouble)与峰值白细胞(WBCPeak)的预测价值。方法 回顾性分析我院血液科收治的78例经亚砷酸诱导治疗的初诊高危APL患者的临床特征,比较WBCDouble和WBCPeak分化综合征的分化程度。结果 分化综合征的发生率为57.69%(45/78),分化综合征最常见的临床症状为发热,重度分化综合征组发热、肺部浸润、心包积液发生率明显高于轻度分化综合征组,差异有统计学意义(P < 0.05)。高危APL患者分化综合征的发生与化疗后WBCPeak(45.21×109/L vs 25.24×109/L,P < 0.001)和化疗后WBCDouble(37.59×109/L vs 17.46×109/L,P=0.007)有关。与WBCPeak组比较,WBCDouble组早于分化事件的发生率高(68.57% vs 40.00%,P=0.016),2组间重度分化、轻度分化和未分化差异均有统计学意义(P=0.002);WBCPeak组出现4个以上分化综合征症状的患者比例更多(28.57% vs 5.71%,P=0.012)。与WBCNO-Double组比较,WBCDouble组患者诊断时白细胞计数(P=0.003)、谷丙转氨酶(P=0.040)、外周血早幼粒细胞数(P=0.047)差异有统计学意义。结论 化疗后WBCDouble和化疗后WBCPeak是分化综合征的危险因素,化疗后WBCDouble较WBCPeak提示分化综合征的发生更有优势。
-
关键词:
- 分化综合征 /
- 高危急性早幼粒细胞白血病 /
- 化疗后WBCDouble /
- 化疗后WBCPeak
Abstract: Objective To screen out the factors that predict the development of differentiation syndrome in new-onset high-risk acute promyelocytic leukemia(APL), and to compare the predictive value of WBCDouble and WBCPeak.Methods The clinical characteristics of 78 newly diagnosed high-risk APL patients treated with arsenious acid as induction therapy were retrospectively analyzed. The differentiation extent of WBCDouble and WBCPeak differentiation syndrome were compared.Results The incidence of differentiation syndrome was 57.69% (45/78). The most common symptom of differentiation syndrome was fever. The incidence of fever, pulmonary infiltration and pericardial effusion in the severe differentiation syndrome group was significantly higher than that in the mild differentiation syndrome group, and there was significant difference (P < 0.05). The development of differentiation syndrome in high-risk APL patients was related to WBCPeak(45.21×109/L vs 25.24×109/L, P < 0.001) and WBCDouble(37.59×109/L vs 17.46×109/L, P=0.007) after chemotherapy. Compared with the WBCPeak group, the incidence of early differentiation events in the WBCDouble group was higher(68.57% vs 40.00%, P=0.016). There were significant differences in severe differentiation, mild differentiation and undifferentiated between the two groups (P=0.002). The WBCPeak group had more than 4 differentiation syndrome symptoms (28.57% vs 5.71%, P=0.012). Compared with the WBCNO-Double group, the white blood cell count (P=0.003), alanine aminotransferase level(P=0.040), and peripheral blood promyelocyte count (P=0.047) at diagnosis in the WBCDouble group were significantly different.Conclusion WBCDouble after chemotherapy and WBCPeak after chemotherapy are risk factors for differentiation syndrome. WBCDouble after chemotherapy can better reflect the development of differentiation syndrome than WBCPeak. -

-
表 1 轻度和重度分化综合征患者的临床表现比较
例(%) 临床症状 轻度(19例) 重度(26例) P 呼吸困难 12(63.16) 19(73.08) 0.483 不明原因的发热 13(68.42) 24(92.31) 0.041 体重增加>5 kg 13(68.42) 22(84.62) 0.202 肺部浸润 9(47.37) 22(84.62) 0.008 心包积液 3(15.79) 12(46.15) 0.035 胸腔积液 8(42.11) 17(65.38) 0.178 低血压 1(5.26) 5(19.23) 0.221 急性肾衰竭 0 5(19.23) 0.125 表 2 分化与未分化患者的临床特征比较
影响因素 未分化组(33例) 分化组(45例) P 性别/例 0.630 男 18 27 女 15 18 年龄/岁 40.33±12.45 44.78±12.55 0.125 Hb/(g/L) 88.33±22.94 78.92±19.52 0.055 PLT/(×109/L) 21.19(14.40) 20.74(24.85) 0.649 WBC/(×109/L) 30.46(25.41) 24.14(31.17) 0.883 WBCPeak/(×109/L) 25.24(39.00) 45.21(51.00) <0.001 WBCDouble/(×109/L) 17.46(42.44) 37.59(36.16) 0.007 ALT/(U/L) 37.10(42.40) 25.20(26.10) 0.504 AST/(U/L) 30.00(27.00) 26.00(20.90) 0.940 MB/% 87.00(13.00) 86.75(11.00) 0.551 PB/% 83.00(25.50) 79.50(23.80) 0.564 Alb/(g/L) 42.30(7.00) 41.10(8.00) 0.123 Cr/(μmol/L) 70.52±15.00 68.16±16.54 0.523 Fib/(g/L) 1.18(1.00) 1.02(1.00) 0.215 UA/(μmol/L) 335.37±111.19 297.20±99.25 0.115 染色体/例 0.896 (15,17易位)及正常核型 26 36 其他 7 9 表 3 WBCDouble和WBCPeak患者的临床症状个数和分化程度比较
例(%) 项目 WBCDouble组(35例) WBCPeak组(35例) P 分化综合征临床表现个数 1 15(42.86) 6(17.14) 0.020 2 12(34.29) 5(14.29) 0.530 3 5(14.29) 9(25.71) 0.235 4 2(5.71) 10(28.57) 0.012 5 1(2.86) 4(11.43) 0.167 6 0 1(2.86) 0.317 分化程度△ 0.002 未分化 15(42.86) 6(17.14) 轻度分化 17(48.57) 14(40.00) 重度分化 3(8.57) 15(42.86) △分化程度定义:≤1个症状诊断为未分化;2~3个症状诊断为轻度分化,≥ 4个症状诊断为重度分化。 表 4 WBCDouble和WBCNO-Double患者的临床特征比较
影响因素 WBCNO-Double (12例) WBCDouble (66例) P 性别/例 0.051 男 10 35 女 2 31 年龄/岁 44.17±11.00 42.67±12.96 0.708 Hb/(g/L) 85.98±20.60 82.35±21.66 0.592 PLT/(×109/L) 26.10(45.20) 21.25(20.19) 0.319 WBC/(×109/L) 53.75(49.87) 26.68(29.54) 0.003 WBCPeak/(×109/L) 22.96(30.00) 40.04(40.00) 0.027 ALT/(U/L) 46.45(45.30) 28.20(31.50) 0.040 AST/(U/L) 44.75(39.50) 29.45(24.70) 0.128 MB/% 87.75(8.00) 87.00(11.00) 0.350 PB/% 86.00(13.30) 81.00(24.60) 0.047 Alb/(g/L) 38.45(8.00) 42.00(6.00) 0.071 Cr/(μmol/L) 77.00±20.97 67.73±14.57 0.063 Fib/(g/L) 1.14(1.00) 1.12(1.00) 0.714 UA/(μmol/L) 308.13± 127.22 314.30± 102.15 0.854 染色体/例 0.976 (15,17易位)及正常核型 9 53 其他 3 13 -
[1] Kulkarni U, Mathews V. Evolving Chemotherapy Free Regimens for Acute Promyelocytic Leukemia[J]. Front Oncol, 2021, 11: 621566. doi: 10.3389/fonc.2021.621566
[2] Nauffal M, Werner L, Ni J, et al. Rate of differentiation syndrome in patients based on timing of initial all-trans retinoic acid administration[J]. Leuk Res Rep, 2019, 12: 100189.
[3] Hou W, Zhang Y, Jin B, et al. Factors affecting thrombohemorrhagic early death in patients with acute promyelocytic leukemia treated with arsenic trioxide alone[J]. Blood Cells Mol Dis, 2019, 79: 102351. doi: 10.1016/j.bcmd.2019.102351
[4] Wen J, Xu F, Zhou Q, et al. Predictors of early death and clinical features in newly diagnosed patients with low-intermediate risk acute promyelocytic leukemia[J]. Front Oncol, 2022, 12: 895777. doi: 10.3389/fonc.2022.895777
[5] Frankel SR, Eardley A, Lauwers G, et al. The 'retinoic acid syndrome' in acute promyelocytic leukemia[J]. Ann Intern Med, 1992, 117: 292-296. doi: 10.7326/0003-4819-117-4-292
[6] Stahl M, Tallman MS. Differentiation syndrome in acute promyelocytic leukaemia[J]. Br J Haematol, 2019, 187(2): 157-162. doi: 10.1111/bjh.16151
[7] 杨柳, 梁晶晶, 董春霞, 等. 急性早幼粒细胞白血病患者合并弥散性血管内凝血的影响因素及列线图模型构建[J]. 临床血液学杂志, 2023, 36(1): 21-26, 32. doi: 10.13201/j.issn.1004-2806.2023.01.005
[8] 中华医学会血液学分会, 中国医师协会血液科医师分会. 中国急性早幼粒细胞白血病诊疗指南(2018年版)[J]. 中华血液学杂志, 2018, 39(3): 179-183.
[9] Danthala M, Golamari KR, Seshachalam A, et al. Walking a Tightrope: Dosage Modifications and Treatment Outcomes of All-Trans-Retinoic Acid, Arsenic Trioxide, and Daunorubicin for High-Risk Acute Promyelocytic Leukemia[J]. JCO Glob Oncol, 2020, 6: 1749-1756.
[10] Sanz MA, Montesinos P, Rayon C, et al. Risk-adapted treatment of acute promyelocytic leukemia based on all-trans retinoic acid and anthracycline with addition of cytarabine in consolidation therapy for high-risk patients: further improvements in treatment outcome[J]. Blood, 2010, 115(25): 5137-5146. doi: 10.1182/blood-2010-01-266007
[11] Sultana J, Dutta J, Mustarin S, et al. Role of Prophylactic Steroids in Differentiation Syndrome[J]. Cureus, 2022, 14(9): e29531.
[12] Gao C, Hu S, Guo M, et al. Clinical pharmacokinetics and safety profile of single agent arsenic trioxide by continuous slow-rate infusion in patients with newly diagnosed acute promyelocytic leukemia[J]. Cancer Chemother Pharmacol, 2018, 82(2): 229-236. doi: 10.1007/s00280-018-3606-8
[13] Yoon JH, Kim HJ, Min GJ, et al. Progressive hyperleukocytosis is a relevant predictive marker for differentiation syndrome, early death, and subsequent relapse in acute promyelocytic leukemia[J]. Sci Rep, 2019, 9(1): 11935. doi: 10.1038/s41598-019-47937-4
[14] Kayser S, Rahme R, Martinez-Cuadron D, et al. Outcome of older(≥70 years)APL patients frontline treated with or without arsenic trioxide-an International Collaborative Study[J]. Leukemia, 2020, 34(9): 2333-2341. doi: 10.1038/s41375-020-0758-4
[15] Wen J, Xu F, Zhou Q, et al. Dynamic changes and multiplication rate of white blood cell count may direct the timing of cytoreduction chemotherapy during induction treatment in newly diagnosed acute promyelocytic leukemia with low-intermediate risk[J]. Mol Clin Oncol, 2021, 14(6): 112. doi: 10.3892/mco.2021.2274
[16] Minamiguchi H, Fujita H, Atsuta Y, et al. Predictors of early death, serious hemorrhage, and differentiation syndrome in Japanese patients with acute promyelocytic leukemia[J]. Ann Hematol, 2020, 99(12): 2787-2800. doi: 10.1007/s00277-020-04245-6
[17] Luo JS, Zhang XL, Huang DP, et al. Differentiation syndrome and coagulation disorder-comparison between treatment with oral and intravenous arsenics in pediatric acute promyelocytic leukemia[J]. Ann Hematol, 2023, 102(7): 1713-1721. doi: 10.1007/s00277-023-05270-x
[18] Kungwankiattichai S, Owattanapanich W, Rattanathammethee T, et al. Does leukocytosis remain a predictive factor for survival outcomes in patients with acute promyelocytic leukemia receiving ATRA plus a chemotherapy-based regimen? A prospective multicenter analysis from TALWG[J]. Hematology, 2023, 28(1): 2191462. doi: 10.1080/16078454.2023.2191462
[19] Gill H, Yung Y, Chu HT, et al. Characteristics and predictors of early hospital deaths in newly diagnosed APL: a 13-year population-wide study[J]. Blood Adv, 2021, 5(14): 2829-2838. doi: 10.1182/bloodadvances.2021004789
[20] Reyhanoglu G, Hughes B, King KE. Differentiation Syndrome, a Side Effect From the Therapy of Acute Promyelocytic Leukemia[J]. Cureus, 2020, 12(12): e12042.
[21] Leblebjian H, DeAngelo DJ, Skirvin JA, et al. Predictive factors for all-trans retinoic acid-related differentiation syndrome in patients with acute promyelocytic leukemia[J]. Leuk Res, 2013, 37(7): 747-751. doi: 10.1016/j.leukres.2013.04.011
[22] Ciftciler R, Haznedaroglu IC, Ozcebe O, et al. The comparison of bone marrow kinetics between patients with acute myeloid leukemia and acute promyelocytic leukemia after induction chemotherapy[J]. Immunopharmacol Immunotoxicol, 2020, 42(1): 17-21. doi: 10.1080/08923973.2019.1692865
-




 下载:
下载: