Effect of different real-time fluorescent quantitative PCR instruments on Epstein-Barr virus DNA detection in peripheral blood
-
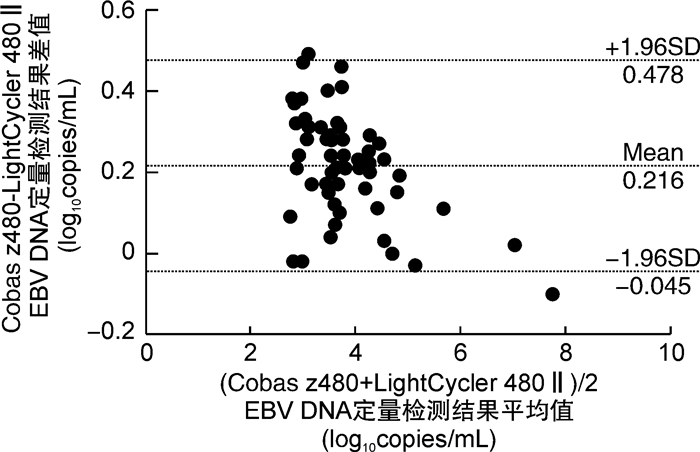
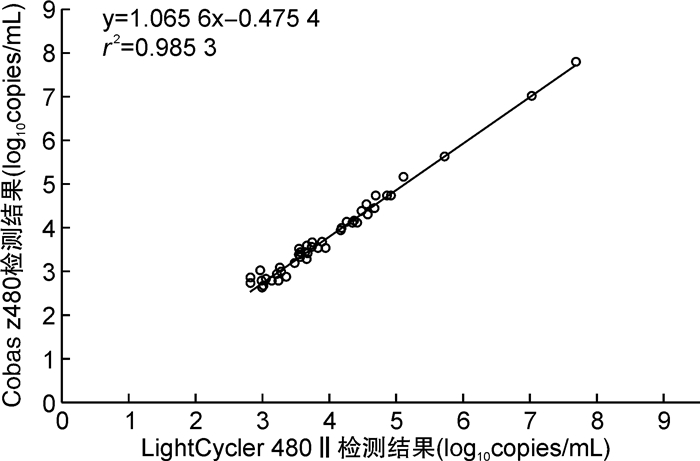
摘要: 目的 应用同一种商品化EB病毒核酸定量检测试剂盒在2款实时荧光定量PCR仪同时检测外周血标本中EB病毒载量,探讨不同类型的实时荧光PCR仪对EBV DNA定性和定量检测结果的影响。方法 提取179例外周血标本(包括102例全血和77例血浆)的DNA后,应用EB病毒核酸定量检测试剂盒(PCR-荧光探针法)分别在2款实时荧光PCR仪Cobas z480和LightCycler 480Ⅱ上同时检测EBV DNA,定性结果采用配对χ2检验和Kappa一致性检验,定量结果采用配对t检验、Spearman相关分析和线性回归分析以及Bland-Altman图分析。结果 ① 应用Cobas z480和LightCycler 480Ⅱ检测179例血标本中EBV DNA的阳性检出率分别为49.72%(89/179)、48.60%(87/179),Kappa值为0.955 3,一致性较好;两者的阳性符合率为98.85%、阴性符合率为96.74%,总符合率为97.77%。②在定量范围内的56例EBV阳性血样本,Cobas z480和LightCycler 480Ⅱ的检测值分别为3.943±0.913、3.726±0.980 log10copies/mL,差异有统计学意义(P< 0.05);进一步进行Spearman相关性以及线性回归分析,相关系数r=0.962 6,回归方程为Y=1.065 6X-0.475 4(r2=0.985 3);2款实时荧光PCR仪检测的差值平均值为0.216 log10copies/mL,差值标准差为0.133 log10copies/mL,95%一致性界限为(-0.045,0.478) log10copies/mL,96.43%(54/56)的点在95%一致性界限以内,界外点数为2(3.57%)。结论 2款实时荧光定量PCR仪Cobas z480和LightCycler 480Ⅱ检测外周血样本中EBV DNA,结果具有可比性,但Cobas z480上的检测结果平均比LightCycler 480Ⅱ上的高,可能是由于2款PCR仪所使用的光源和滤光片组合不同所致。
-
关键词:
- EB病毒 /
- 定量 /
- 实时荧光定量PCR仪
Abstract: Objective To compare the results of peripheral blood clinical specimens obtained with a commercial Epstein-Barr virus(EBV) DNA quantitative fluorescence diagnostic kit(PCR-fluorescence probing) using two platforms: Cobas z480 real-time PCR and LightCycler 480Ⅱreal-time PCR detection systems, and determine the concordance on qualitative and quantitative results of EBV DNA obtained by these two different real-time PCR instruments.Methods Nucleic acid was extracted from 102 whole blood(WB) and 77 plasma clinical specimens using Sansure commercial extraction kit. Real-time PCR was performed on both Cobas z480 and LightCycler 480Ⅱreal-time PCR platforms, using Sansure EBV DNA quantitative fluorescence diagnostic kit. Comparison of the results obtained with two types of thermocyclers included Cohen's kappa, linear regression analysis, pairedttest, Spearman correlation, and Bland-Altman analysis.Results ① The detection rates of 179 peripheral blood clinical samples obtained by Cobas z480 and LightCycler 480Ⅱplatforms were 49.72%(89/179) and 48.60%(87/179) respectively. The excellent agreement was observed between both platforms(κ=0.955 3). The positive coincidence rate, negative coincidence rate and overall coincidence rate were 98.85%, 96.74% and 97.77%, respectively. ②A total of 56 clinical specimens with EBV DNA load above the limit of quantification on both PCR platforms were analyzed. The detection values of EBV DNA load obtained by Cobas z480 and LightCycler 480Ⅱplatforms were 3.943±0.913 and 3.726±0.980 log10copies/mL, respectively(P< 0.05). Correlation coefficient of the results obtained with both analyzers was 0.962 6. Bland-Altman plot analysis found the difference of mean values measured with both PCR thermocyclers was 0.216 log10copies/mL(95% confidence interval, -0.045 to 0.478), and the proportions of the specimens within the 95% limit of agreement were 96.43%(54/56) between both platforms.Conclusion The two real-time fluorescent PCR instruments provide reliable and comparable EBV viral load results, but viral load values measured with the Cobas z480 platform are on average higher than those measured with LightCycler 480Ⅱplatform. The difference is probably due to different light sources and filters used in both thermocyclers. -

-
表 1 2款实时荧光PCR仪定性检测血标本中EBV DNA的结果分析
Cobas z480 LightCycler 480Ⅱ Kappa
(95%CI)阳性符合率/%
(95%CI)阴性符合率/%
(95%CI)总符合率/%
(95%CI)阳性 阴性 阳性
阴性86
13
890.9553(0.9120~
0.9986)98.85(93.76~
99.97)96.74(90.76~
99.32)97.77(94.38
~99.39)表 2 2款实时荧光PCR仪的常用染料的激发光谱和发射光谱
nm 染料 Cobas z480 LightCycler 480Ⅱ λ激发
滤光片λ发射
滤光片λ激发
滤光片λ发射
滤光片FAM 465 510 440 510 HEX/VIC 540 580 533 580 Red610 498 610 498 610 Cy5 610 670 618 660 -
[1] Kanakry JA, Hegde AM, Durand CM, et al. The clinical significance of EBV DNA in the plasma and peripheral blood mononuclear cells of patients with or without EBV diseases[J]. Blood, 2016, 127(16): 2007-2017. doi: 10.1182/blood-2015-09-672030
[2] Chan K, Woo J, King A, et al. Analysis of Plasma Epstein-Barr Virus DNA to Screen for Nasopharyngeal Cancer[J]. N Engl J Med, 2017, 377(6): 513-522. doi: 10.1056/NEJMoa1701717
[3] Hayden RT, Yan X, Wick MT, et al. Factors contributing to variability of quantitative viral PCR results in proficiency testing samples: a multivariate analysis[J]. J Clin Microbiol, 2012, 50(2): 337-345. doi: 10.1128/JCM.01287-11
[4] Pavši J, Devonshire AS, Parkes H, et al. Standardization of Nucleic Acid Tests for Clinical Measurements of Bacteria and Viruses[J]. J Clin Microbiol, 2015, 53(7): 2008-2014. doi: 10.1128/JCM.02136-14
[5] 耿帜, 周志明, 肖圣达, 等. 两款荧光定量PCR仪检测HLA-B27基因结果比对分析[J]. 临床血液学杂志, 2019, 32(8): 593-596. http://lcxz.cbpt.cnki.net/WKC/WebPublication/paperDigest.aspx?paperID=518d86db-9a9c-4b28-9359-88aff8855766
[6] Przybylski M, Dzieciatkowski T, Les K, et al. Comparison of real-time PCR quantitative analysis of the cytomegalovirus DNA level using LightCycler 2.0 and LightCycler 480 instruments[J]. J Clin Virol, 2012, 55(3): 270-273. doi: 10.1016/j.jcv.2012.08.005
-




 下载:
下载: