Clinical observation of consolidation chemotherapy combined with allogeneic NK cells infusion in the treatment of elderly acute myeloid leukemia
-
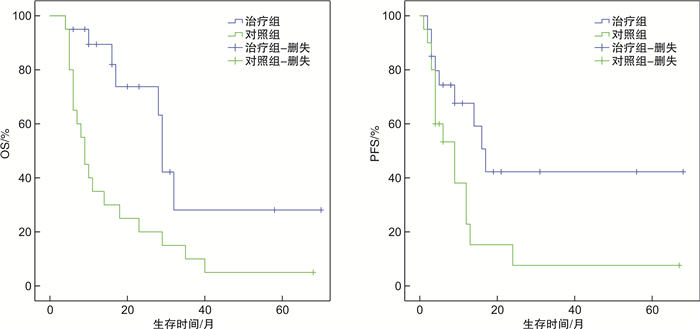
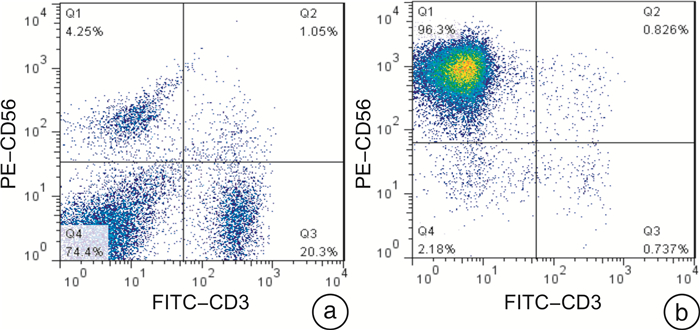
摘要: 目的 探讨巩固化疗联合异基因NK细胞输注治疗老年急性髓系白血病(AML)的疗效及安全性。方法 对2016年8月—2022年1月20例老年AML完全缓解(CR)患者(治疗组),给予巩固化疗联合异基因NK细胞输注2~6个疗程,平均每例4个疗程,观察患者的安全性、分子生物学缓解率及复发率。并与20例仅接受巩固化疗的老年AML CR患者(对照组)进行比较,观察2组间中位生存时间、无复发生存(RFS)率、总生存(OS)率的差异。结果 治疗组20例患者共接受巩固化疗联合NK细胞输注79次,输注NK细胞数量(7.61±2.50)×107/(kg·次)。18例第1次CR(CR1)患者中14例(77.78%)获得分子生物学CR,其中仅2例复发,4例未获得分子生物学CR者均出现复发,总复发率33.33%(6/18);2例第2次CR(CR2)患者均未获得分子生物学CR,其中1例再次复发,1例死于心功能不全。对照组复发率75.00%(15/20),仅1例长期生存。治疗组中位生存时间29个月(95%CI27.58~30.43),对照组中位生存时间9个月(95%CI6.82~11.18)。治疗组1年、2年、3年OS率分别为(89.4±7.1)%、(73.8±11.7)%、(28.1±15.4)%,对照组分别为(35.0±10.7)%、(20.0±8.9)%、(10.0±6.7)%,2组比较差异有统计学意义(P=0.003)。治疗组1年、2年、3年RFS率分别为(67.6±11.0)%、(42.3±13.5)%、(42.3±13.5)%,对照组分别为(22.9±11.1)%、(7.6±7.2)%、(7.6±7.2)%,2组比较差异有统计学意义(P=0.029)。治疗组79次NK细胞输注中,7次输注出现短暂低至中度发热,输注期间及输注后无其他不良反应发生。结论 巩固化疗联合异基因NK细胞输注治疗老年AML是安全的,可能使更多老年AML患者获得分子生物学CR,降低复发率,提高中位生存期、OS率及RFS率,为老年AML的治疗提供了新的选择。Abstract: Objective To investigate the efficacy and safety of consolidation chemotherapy combined with allogeneic NK cells infusion in the treatment of elderly patients with acute myeloid leukemia(AML).Methods From August 2016 to January 2022, 20 elderly patients with AML at the stage of complete remission(CR) were treated with 2 to 6 courses of consolidation chemotherapy combined with allogeneic NK cells infusion(treatment group), with an average of 4 courses per case. The safety, molecular remission rate and recurrence rate were observed. Compared with 20 cases of elderly patients with AML CR who had received consolidation chemotherapy only(control group), the differences in median survival time, relapse-free survival rate(RFS) and overall survival rate(OS) between the two groups were analyzed.Results In the treatment group, 20 patients received 79 times of consolidation chemotherapy combined with NK cells infusion. The doses of infused NK cells were(7.61±2.50) ×107/kg per infusion. Among the 18 patients with the first CR(CR1), 14 cases (77.78%) achieved molecular biological CR, of whic only 2 cases relapsed, but all 4 patients without molecular biological CR relapsed, the overall recurrence rate was 33.33%(6/18). Two patients with the second CR(CR2) did not achieve molecular biological CR, and one case relapsed again, the other one died of cardiac dysfunction. In the control group, the recurrence rate was 75.00%(15/20), only one patient obtained long-term survival. The median survival time was 29 months(95%CI27.58-30.43) in the treatment group and 9 months(95%CI6.82-11.18) in the control group. The 1-year, 2-year and 3-year OS were(89.4±7.1)%, (73.8±11.7)%, (28.1±15.4)% respectively in the treatment group, and(35.0±10.7)%, (20.0±8.9)%, (10.0±6.7)% respectively in the control group, there was significant difference between 2 groups(P=0.003). The 1-year, 2-year and 3-year RFS were(67.6±11.0)%, (42.3±13.5)%, (42.3±13.5)% respectively in the treatment group, and(22.9±11.1)%, (7.6±7.2)%, (7.6±7.2)% respectively in the control group, there was significant difference between 2 groups(P=0.029). Among 79 times of NK cells infusion in the treatment group, transient low to moderate fever occurred in 7 times of infusion. No other toxicities occurred during and after infusion.Conclusion Consolidation chemotherapy combined with allogeneic NK cells infusion is safe and may enable more elderly AML patients to obtain molecular biological CR, reduce recurrence rate, improve median survival time, OS and RFS. It provides a new treatment option for elderly AML.
-
Key words:
- acute myelocytic leukemia /
- natural killer cells /
- immunotherapy /
- chemotherapy
-

-
[1] 张伟, 方芳, 何颖, 等. HCT-CI评分指导老年性急性髓系白血病治疗选择[J]. 中国实验血液学杂志, 2017, 25(2): 387-392. https://www.cnki.com.cn/Article/CJFDTOTAL-XYSY201702017.htm
[2] 战榕. Bcl-2抑制剂在急性髓系白血病中的研究进展: 第62届美国血液学年会报道[J]. 临床血液学杂志, 2021, 34(5): 298-301. doi: 10.13201/j.issn.1004-2806.2021.05.002
[3] 郭怀鹏, 康蕾, 刘聪, 等. 去甲基化药物治疗老年急性髓系白血病的Meta分析[J]. 临床血液学杂志, 2021, 34(5): 347-352. doi: 10.13201/j.issn.1004-2806.2021.05.011
[4] Knorr DA, Bachanova V, Verneris MR, et al. Clinical utility of natural killer cell in cancer therapy and transplantation[J]. Semin Immunol, 2014, 26(2): 161-172. doi: 10.1016/j.smim.2014.02.002
[5] 沈悌, 赵永强. 血液病诊断及疗效标准[M]. 4版. 北京: 科学出版社, 2018: 91-96.
[6] Schuurhuis GJ, Heuser M, Freeman S, et al. Minimal/measurable residual disease in AML: a consensus document from the Eumpean Leukemia Net MRD Working Party[J]. Blood, 2018, 131(12): 1275-1291. doi: 10.1182/blood-2017-09-801498
[7] Zhang Y, Huang B. The development and diversity of ILCs, NK cells and their relevance in health and diseases[J]. Adv Exp Med Biol, 2017, 10(24): 225-244.
[8] Jeffrey S, Miller I, Lewis L. Natural killer cells in cancer immunotherapy[J]. Ann Rev Cancer Biol, 2019, 3(1): 77-103. doi: 10.1146/annurev-cancerbio-030518-055653
[9] Hu W, Wang G, Huang D, et al. Cancer Immunotherapy Based on Natural Killer Cells: Current Progress and New Opportunities[J]. Front Immuno, 2019, 10: 1205-1220. doi: 10.3389/fimmu.2019.01205
[10] Sanchez CE, Dowlati EP, Geiger AE, et al. NK Cell Adoptive Immunotherapy of Cancer: Evaluating Recognition Strategies and Overcoming Limitations[J]. Transplant Cell Ther, 2021, 27(1): 21-35. doi: 10.1016/j.bbmt.2020.09.030
[11] Lupo KB, Matosevic S. Natural Killer Cells as Allogeneic Effectors in Adoptive Cancer Immunotherapy[J]. Cancers, 2019, 11(6): 769-793. doi: 10.3390/cancers11060769
[12] Veluchamy JP, Kok N, van der Vliet HJ, et al. The Rise of Allogeneic Natural Killer Cells as a Platform for Cancer Immunotherapy: Recent Innovations and Future Developments[J]. Front Immunol, 2017, 8: 631-620. doi: 10.3389/fimmu.2017.00631
[13] Fehniger TA, Miller JS, Stuart RK, et al. A Phase 1 Trial of CNDO-109-Activated Natural Killer Cells in Patients with High-Risk Acute Myeloid Leukemia[J]. Biol Blood Marrow Transplant, 2018, 24(8): 1581-1589. doi: 10.1016/j.bbmt.2018.03.019
[14] Hattori N, Nakamaki T. Natural Killer Immunotherapy for Minimal Residual Disease Eradication Following Allogeneic Hematopoietic Stem Cell Transplantation in Acute Myeloid Leukemia[J]. Int J Mol Sci, 2019, 20(9): 2057-2073. doi: 10.3390/ijms20092057
[15] Muñoz Builes M, Vela Cuenca M, Fuster Soler JL, et al. Study protocol for a phase Ⅱ, multicentre, prospective, non-randomised clinical trial to assess the safety and efficacy of infusing allogeneic activated and expanded natural killer cells as consolidation therapy for paediatric acute myeloblastic leukaemia[J]. BMJ Open, 2020, 10(1): e029642. doi: 10.1136/bmjopen-2019-029642
[16] 王春键, 黄晓军, 宫立众, 等. 化疗联合异基因自然杀伤细胞巩固治疗低中危急性髓系白血病的疗效观察[J]. 中华血液学杂志, 2019, 40(10): 812-817. doi: 10.3760/cma.j.issn.0253-2727.2019.10.003
[17] Curti A, Ruggeri L, D'Addio A, et al. Successful transfer of alloreactive haploidentical KIR ligand-mismatched natural killer cells after infusion in elderly high risk acute myeloid leukemia patients[J]. Blood, 2011, 118(12): 2373-3279.
[18] Curti A, Ruggeri L, Parisi S, et al. Larger size of donor alloreactive NK Cell repertoire correlates with better response to NK cell immunotherapy in elderly acute myeloid leukemia patients[J]. Clin Cancer Res, 2016, 22(8): 1914-1921. doi: 10.1158/1078-0432.CCR-15-1604
[19] Gonzalez-Rodriguez AP, Villa-Álvarez M, Sordo-Bahamonde C, et al. NK Cells in the Treatment of Hematological Malignancies[J]. J Clin Med, 2019, 8(10): 1557-1579. doi: 10.3390/jcm8101557
[20] Dolstra H, Roeven MWH, Spanholtz J, et al. Successful Transfer of Umbilical Cord Blood CD34+ Hematopoietic Stem and Progenitor-derived NK Cells in Older Acute Myeloid Leukemia Patients[J]. Clin Cancer Res, 2017, 23(15): 4107-4118. doi: 10.1158/1078-0432.CCR-16-2981
-




 下载:
下载: