Study on stability of M, Fya and P1 antigens in antibody screening cells within validity period
-
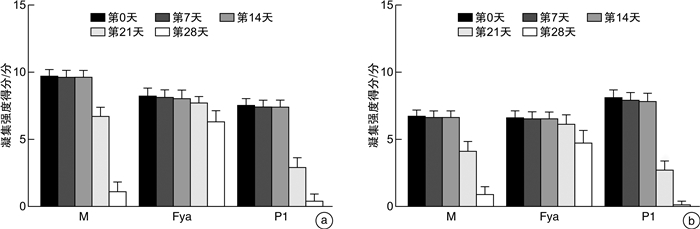
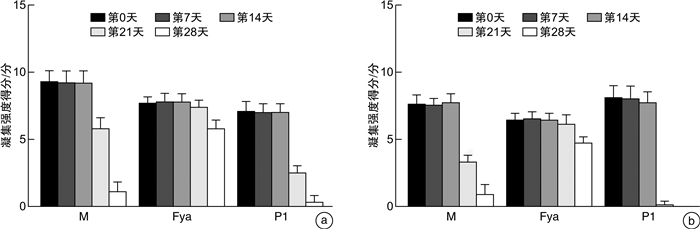
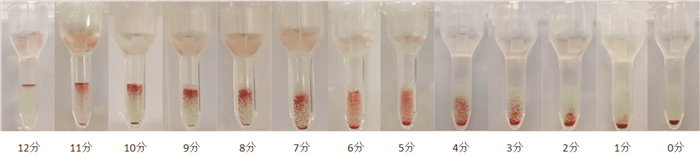
摘要: 目的研究抗体筛查细胞在保存期内M抗原、Fya抗原和P1抗原的稳定性变化。方法应用2个不同厂家的抗体筛查细胞通过倍比稀释法进行单克隆抗体标化(IgG-M/Fya/P1抗体)。收集抗-M/Fya/ P1抗体阳性标本,制备人源抗体血清。自配1%试剂红细胞(10例O型且M/Fya/P1抗原阳性志愿者)验证标化单克隆抗体和人源抗体的稳定性。在有效期最后30 d内,采用微柱凝集法,使用标化单克隆抗体和人源抗体对2厂家抗体筛查细胞(试剂1和2) M/Fya/P1抗原的抗原性进行监测。结果试剂1,M和P1抗原在第21天及Fya抗原在第28天与第0天比较,凝聚强度结果差异有统计学意义(P<0.05);M抗原于第28天及P1抗原于第21天出现无法检出现象。试剂2,M和P1抗原在第21天及Fya抗原在第28天出现凝聚强度差异(P<0.05);M和P1抗原第28天出现无法检出现象。结论抗体筛查细胞血型抗原稳定性在有效期内逐渐降低,建议抗体筛查细胞保存期间进行质量监测。Abstract: ObjectiveTo study the stability changes of M, Fya and P1 antigens in antibody screening cells during the storage period.MethodsMonoclonal antibody standardization(IgG-M/Fya/P1 antibody) was performed by doubling dilution using antibody screening cells from two different manufacturers. The positive specimens of anti-M/Fya/P1 antibody in our department were collected to prepare human antibody serum. Self-prepared 1% reagent red blood cells(10 volunteers with O type and positive for M/Fya/P1 antigen) were used to verify the stability of standardized monoclonal antibodies and human antibodies. During the last 30 days of validity, the M/Fya/P1 antigen antigenicity of the two manufacturers' antibody screening cells(Reagent 1 and Reagent 2) was monitored by microcolumn agglutination and using standardized monoclonal antibodies and human antibodies.ResultsFor reagent 1, compared with day 0, the results of aggregation strength of M and P1 antigens on day 21 and Fya antigen on day 28 were significantly different(P < 0.05). The M antigen was undetectable on the 28th day and the P1 antigen on the 21st day. For reagent 2, the aggregation intensity of M and P1 antigens was different on the 21st day and the Fya antigen on the 28th day(P < 0.05). The M and P1 antigens were undetectable on day 28.ConclusionThe stability of antibody screening cells blood group antigen gradually decreased during the validity period. It is recommended to monitor the quality of antibody screening cells during storage.
-
Key words:
- antibody screening cells /
- stability /
- column agglutination /
- human antibody /
- monoclonal antibody
-

-
表 1 3种单克抗体在标化稀释倍数表
抗体名称 试剂1 试剂2 单克隆抗体IgG-M 16 10 单克隆抗体IgG-P1 4 2 单克隆抗体IgG-Fya 10 5 表 2 标化单克隆抗体和人源抗体保存稳定性验证
n=10,X±S 验证时间 标化单克隆抗体 人源抗体 试剂1 试剂2 M抗原 Fya抗原 P1抗原 M抗原 Fya抗原 P1抗原 M抗原 Fya抗原 P1抗原 第0天 9.200±0.789 8.200±0.789 8.500±0.707 8.700±0.483 8.400±0.699 8.900±0.539 8.500±0.527 8.600±0.516 8.500±0.527 第30天 9.100±0.737 8.300±0.823 8.600±0.789 8.600±0.516 8.400±0.843 8.600±0.490 8.600±0.517 8.400±0.516 8.700±0.483 第60天 9.300±0.675 8.100±0.738 8.400±0.699 8.800±0.421 8.300±0.823 8.700±0.640 8.600±0.700 8.500±0.527 8.500±0.527 F 0.310 0 0.980 0 0.560 0 1.590 0 0.180 0 1.190 0 0.100 0 0.310 0 0.550 0 P 0.737 0 0.393 0 0.582 2 0.231 6 0.833 7 0.327 4 0.904 3 0.737 0 0.588 9 表 3 试剂1的M、P1和Fya抗原稳定性监测表
n=10,X±S 验证时间 单克隆抗体 人源抗体 M抗原 Fya抗原 P1抗原 M抗原 Fya抗原 P1抗原 第0天 9.700±0.483 8.200±0.632 7.500±0.527 6.700±0.483 6.600±0.516 8.100±0.568 第7天 9.600±0.516 8.100±0.568 7.400±0.516 6.600±0.516 6.500±0.527 7.900±0.568 第14天 9.600±0.516 8.000±0.667 7.400±0.516 6.600±0.516 6.500±0.527 7.800±0.632 第21天 6.700±0.675 7.700±0.483 2.900±0.738 4.100±0.738 6.100±0.738 2.700±0.675 第28天 1.100±0.738 6.300±0.823 0.400±0.516 0.900±0.567 4.700±0.949 0.100±0.316 F 368.090 22.340 356.600 331.310 18.470 960.610 P <0.001 <0.001 <0.001 <0.001 <0.001 <0.001 表 4 试剂2的M、P1和Fya抗原稳定性监测表
n=10,X±S 时间 单克隆抗体 人源抗体 M抗原 Fya抗原 P1抗原 M抗原 Fya抗原 P1抗原 第0天 9.300±0.823 7.700±0.483 7.100±0.738 7.600±0.699 6.400±0.516 8.100±0.876 第7天 9.200±0.919 7.800±0.632 7.000±0.667 7.500±0.527 6.500±0.527 8.000±0.943 第14天 9.200±0.919 7.800±0.632 7.000±0.667 7.700±0.675 6.400±0.516 7.700±0.823 第21天 5.800±0.789 7.400±0.516 2.500±0.527 3.300±0.483 6.100±0.738 0.100±0.316 第28天 1.100±0.738 5.800±0.632 0.300±0.483 0.900±0.738 4.700±0.483 0 F 212.100 38.650 408.710 490.000 38.370 428.910 P <0.001 <0.001 <0.001 <0.001 <0.001 <0.001 -
[1] 杨东, 陈青. 红细胞血型系统研究新进展[J]. 临床输血与检验, 2022, 24(2): 137-141, 146. doi: 10.3969/j.issn.1671-2587.2022.02.001
[2] 仲照东, 陈智超, 夏凌辉. 急性髓系白血病造血干细胞移植后复发的预防及治疗[J]. 临床血液学杂志, 2022, 35(5): 375-379. http://lcxz.cbpt.cnki.net/WKC/WebPublication/paperDigest.aspx?paperID=a1c52ff4-78ab-41c5-96f3-d9824f5c4941
[3] 乔静, 刘庆生, 庞新丰, 等. 53例Rh血型系统以外不规则抗体分布及新生儿溶血病发生关联分析[J]. 中国输血杂志, 2021, 34(8): 874-877. https://www.cnki.com.cn/Article/CJFDTOTAL-BLOO202108019.htm
[4] 杨婷婷. 输血前不规则抗体筛查与输血安全的重要作用研究[J]. 中国继续医学教育, 2021, 13(33): 134-137. doi: 10.3969/j.issn.1674-9308.2021.33.036
[5] 李洪兵, 王纯, 文玲, 等. 不规则抗体检测对非Rh血型系统患者输血不良反应及溶血的影响[J]. 临床和实验医学杂志, 2021, 20(20): 2215-2218. doi: 10.3969/j.issn.1671-4695.2021.20.025
[6] 张黎雯, 李树中, 田丰, 等. 红细胞血型抗原的最新研究进展[J]. 临床血液学杂志, 2021, 34(2): 135-144. http://lcxz.cbpt.cnki.net/WKC/WebPublication/paperDigest.aspx?paperID=6621d555-9660-4750-bbe6-59e41ef21862
[7] Ciana A, Achilli C, Gaur A, et al. Membrane Remodelling and Vesicle Formation During Ageing of Human Red Blood Cells[J]. Cell Physiol Biochem, 2017, 42(3): 1127-1138. doi: 10.1159/000478768
[8] Lee HS, Choi KM, Won EJ, et al. Protein stability changes of the novel p. Arg180Cys mutant A glycosyltransferase resulted in a weak A phenotype[J]. Vox Sang, 2016, 111(4): 441-444. doi: 10.1111/vox.12440
[9] 赵俸涌, 李勤, 郭忠慧, 等. 保存期间试剂红细胞血型相关膜蛋白抗原性研究[J]. 中国输血杂志, 2018, 31(9): 957-959. https://www.cnki.com.cn/Article/CJFDTOTAL-BLOO201809015.htm
[10] Remigante A, Morabito R, Marino A. Natural Antioxidants Beneficial Effects on Anion Exchange through Band 3 Protein in Human Erythrocytes[J]. Antioxidants(Basel), 2019, 9(1): 25.
[11] 方鹏, 李玲, 何芮, 等. 红细胞膜表面黏附分子与红细胞寿命的相关性研究[J]. 中国输血杂志, 2021, 34(1): 19-22. https://www.cnki.com.cn/Article/CJFDTOTAL-BLOO202101008.htm
[12] 柳斌, 王伟, 樊瑞军, 等. 血型不规则抗体筛查在精准输血管理中的应用[J]. 宁夏医学杂志, 2021, 43(2): 139-141. https://www.cnki.com.cn/Article/CJFDTOTAL-NXYX202102018.htm
[13] 赵震, 孙长杰, 王晓宁, 等. 抗人球蛋白IgG和C_3d对不规则抗体筛查项目诊断价值分析[J]. 临床血液学杂志, 2020, 33(2): 101-104. http://lcxz.cbpt.cnki.net/WKC/WebPublication/paperDigest.aspx?paperID=2ef8851d-1c05-4674-9723-d32fdb735270
[14] 刘世佳, 张勇萍, 杨世明, 等. IgM抗Le~a、抗P_1抗体联合IgG抗E抗体的血清学检测分析[J]. 细胞与分子免疫学杂志, 2021, 37(10): 932-935. https://www.cnki.com.cn/Article/CJFDTOTAL-XBFM202110011.htm
[15] 钱明明, 郭霞, 乔芳, 等. 母婴ABO血型不合合并IgG抗-M抗体引起新生儿溶血病1例[J]. 安徽医药, 2021, 25(10): 2102-2105. https://www.cnki.com.cn/Article/CJFDTOTAL-AHYY202110045.htm
-




 下载:
下载: