Study on correlation between chronic mountain sickness and blood routine and blood biochemical indexes
-
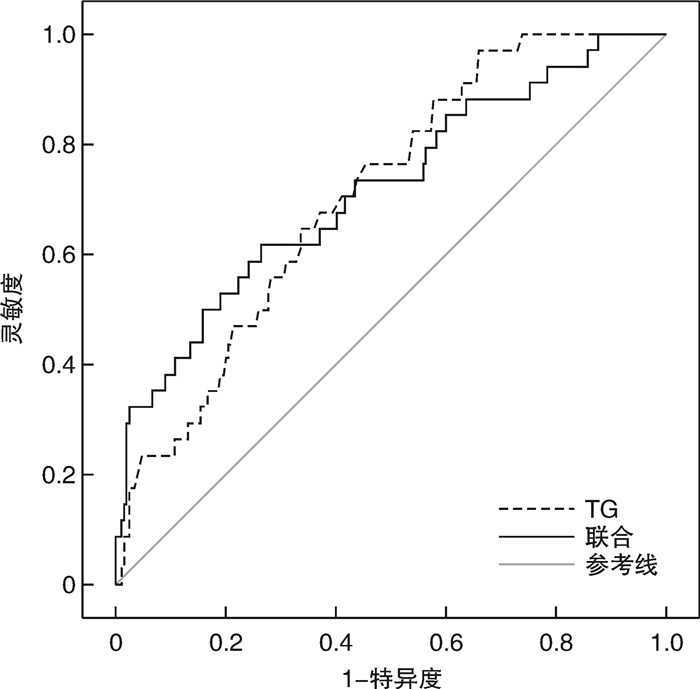
摘要: 目的 检测久居高海拔地区人群血常规和血生化指标情况,并探讨其与慢性高原病(CMS)之间的关系,为常驻高原人群健康研究提供参考。方法 选取在2 880~5 400 m海拔生活1个月以上的252例男性受试者为研究对象,对受试者静脉血进行血常规和血生化指标检测,并通过血细胞分析参考区间和临床常用生化检验项目参考区间判断血常规和血生化指标异常情况;应用CMS量表调查CMS患病情况,并比较血常规和血生化指标在CMS患病人群和非患病人群之间的差异;分析血常规及血生化指标与CMS的关系。结果 ① 多项血液指标在常驻高原人群中存在异常,其中红细胞数量、血红蛋白浓度(HGB)、红细胞压积(HCT)、平均血小板体积以及尿酸(UA)水平高于正常参考值上限的人群分别占比71.0%、75.8%、96.4%、82.1%和72.1%;血小板分布宽度低于正常参考值下限的人群占比76.3%。②与非CMS患病人群相比,CMS患病人群的HGB、HCT、总蛋白、甘油三酯(TG)和低密度脂蛋白胆固醇水平显著增高;③与非CMS患病人群相比,CMS患病人群的血小板计量和总胆汁酸水平的异常率显著增高;④嗜酸性粒细胞百分比增加(EOS%)、HGB升高和TG增高是CMS患病的主要危险因素;⑤TG的受试者工作特征曲线(ROC曲线)预测CMS的ROC曲线下的面积(AUC)为0.710。结论 EOS%、HGB和TG增高是CMS患病的主要危险因素,其中TG水平增高可用于CMS早期预警,值得重点关注。Abstract: Objective To detect the blood routine and blood biochemical indexes of residents in plateau areas, and analyze their association with chronic mountain sickness(CMS), providing a reference for the health research of the plateau population.Methods A total of 252 male subjects living at an altitude of 2 880-5 400 m for more than one month were selected as the research subjects. The blood routine and blood biochemical indexes were detected, and the abnormal blood routine and blood biochemical indexes were judged by the reference intervals for blood cell analysis and the reference intervals for common clinical biochemistry tests. The CMS scale was used to investigate the prevalence of CMS, and the differences between blood routine and blood biochemical indexes in the CMS patients and non-CMS people were compared. The relationship between blood routine and blood biochemical indexes and CMS was analyzed.Results ① Multiple blood indicators were abnormal in the residents in plateau areas, among which the number of red blood cells, hemoglobin concentration(HGB), hematocrit, mean platelet volume and uric acid levels were higher than the upper limit of normal reference values accounted for 71.0%, 75.8%, 96.4%, 82.1% and 72.1%, respectively; the width of platelet distribution was lower than the lower limit of the normal reference value in 76.3% of the population. ②The levels of hemoglobin concentration, hematocrit, total protein, triglycerides, and LDL cholesterol in CMS patients were significantly increased.③The abnormal rate of platelet number and total bile acid level in CMS patients was significantly increased.④Eosinophil percentage, hemoglobin concentration, and triglycerides were the main risk factors of CMS. ⑤The receiver operating characteristic curve of TG revealed that the AUC of CMS prediction was 0.710.Conclusion EOS%, HGB, and TG were the main risk factors of CMS, and TG level changes could be used for early warning of CMS, which deserved special attention.
-
Key words:
- plateau /
- hypoxia /
- chronic mountain sickness /
- blood routine /
- blood biochemistry
-

-
表 1 非CMS患者和CMS患者血液指标比较
X±S 指标 非CMS患者(n=185) CMS患者(n=67) HGB/(g/L) 187.06±16.82 199.85±25.282) HCT/% 57.13±4.66 60.93±8.091) TP/(g/L) 70.84±3.92 72.47±5.591) TG/(mmol/L) 1.04±0.52 1.41±0.612) LDL-C/(mmol/L) 2.23±0.63 2.50±0.631) 与非CMS患者比较,1)P < 0.05,2)P < 0.01。 表 2 非CMS患者和CMS患者血液指标异常率比较
指标 指标异常率/% χ2 P 非CMS患者 CMS患者 PLT 2.80 14.70 10.068 < 0.01 TBA 9.20 20.60 3.960 < 0.05 表 3 CMS患病危险因素单因素回归分析结果
项目 回归系数 SE Wald OR 95%CI P EOS% 0.251 0.115 4.786 1.286 1.027~1.610 0.029 RBC 0.558 0.256 4.752 1.746 1.058~2.883 0.029 HGB 0.032 0.009 12.296 1.033 1.014~1.052 < 0.001 HCT 0.113 0.032 12.570 1.120 1.052~1.192 < 0.001 MCH 0.251 0.123 4.179 1.285 1.010~1.635 0.041 TP 0.084 0.041 4.192 1.087 1.004~1.178 0.041 TG 1.002 0.297 11.359 2.724 1.521~4.878 0.001 CHOL 0.494 0.228 4.681 1.638 1.048~2.563 0.030 LDL-C 0.628 0.279 5.065 1.874 1.084~3.239 0.024 表 4 CMS患病危险因素多因素回归分析结果
项目 回归系数 SE Wald OR 95%CI P EOS% 0.335 0.123 7.456 1.398 1.009~1.777 < 0.01 HGB 0.031 0.010 9.701 1.032 1.012~1.052 < 0.01 TG 0.754 0.309 5.973 2.126 1.161~3.892 < 0.05 常数 -9.352 2.003 21.799 表 5 TG及EOS%、HGB、TG联合分析对CMS的预测价值
指标 AUC 94%CI 截断值/(mmol/L) 灵敏度 特异度 P TG 0.710 0.627~0.793 0.955 0.765 0.548 < 0.001 EOS%、HGB、TG联合 0.718 0.620~0.817 - 0.618 0.737 < 0.001 -
[1] Rimoldi SF, Rexhaj E, Pratali L, et al. Systemic vascular dysfunction in patients with chronic mountain sickness[J]. Chest, 2012, 141(1): 139-146. doi: 10.1378/chest.11-0342
[2] 王红. 血常规检测的临床意义分析与研究[J]. 青岛医药卫生, 2013, 45(2): 140-141. https://www.cnki.com.cn/Article/CJFDTOTAL-QDYW201302029.htm
[3] Yang W, Zhao S, Liu D, et al. Establishment of reference intervals for blood cell analysis of adult Tibetan farmers and herdsmen over 4100 meters above sea level in Tibet based on a health survey[J]. High Alt Med Biol, 2020, 21(3): 223-231. doi: 10.1089/ham.2020.0006
[4] 宋佳琪, 马振, 曾泽, 等. 某部长期驻4500 m高原官兵血常规及血生化检查特点分析[J]. 人民军医, 2021, 64(9): 801-804. https://www.cnki.com.cn/Article/CJFDTOTAL-RMJZ202109001.htm
[5] 中华医学会高原医学分会. 关于统一使用慢性高原(山)病"青海标准"的决定[J]. 高原医学杂志, 2007, 17(1): 1-2. https://www.cnki.com.cn/Article/CJFDTOTAL-GYYZ200701000.htm
[6] 牛园满, 李振宇, 邱婷婷, 等. 外周血中性粒细胞/淋巴细胞比值与真性红细胞增多症血栓事件关系的研究[J]. 临床血液学杂志, 2021, 34(3): 192-197. https://www.cnki.com.cn/Article/CJFDTOTAL-LCXZ202103011.htm
[7] 卢瑞, 张梦潇, 张爱边, 等. 红细胞分布宽度对原发性血小板增多症和真性红细胞增多症患者血栓形成及其预后的预测价值[J]. 临床血液学杂志, 2021, 34(11): 785-789. https://lcxy.whuhzzs.com/article/doi/10.13201/j.issn.1004-2806.2021.11.008
[8] 余雯静, 王大新. 急性感染诱发急性心肌梗死的临床特点及炎症指标相关分析[J]. 临床心血管病杂志, 2021, 37(7): 615-619.
[9] 王俊力, 鄢翎, 伍燕, 等. 高原地区慢性阻塞性肺疾病患者肺动脉高压发生率及危险因素分析[J]. 检验医学与临床, 2018, 15(12): 1803-1805, 1868. https://www.cnki.com.cn/Article/CJFDTOTAL-JYYL201812032.htm
[10] 陈火元, 朱鲜阳, 王琦光, 等. 平均血小板体积与动脉型肺动脉高压关系的研究[J]. 生物医学工程与临床, 2018, 22(2): 183-186. https://www.cnki.com.cn/Article/CJFDTOTAL-SGLC201802013.htm
[11] Mäntysaari M, Joutsi-Korhonen L, Siimes MA, et al. Unaltered blood coagulation and platelet function in healthy subjects exposed to acute hypoxia[J]. Aviat Space Environ Med, 2011, 82(7): 699-703.
[12] Tyagi T, Ahmad S, Gupta N, et al. Altered expression of platelet proteins and calpai activity mediate hypoxia-induced prothrombotic phenotype[J]. Blood, 2014, 123(8): 1250-1260.
[13] McDonald TP, Cottrell MB, Swearingen CJ, et al. Comparative effects of thrombopoietin and interleukin-6 on murine megakaryocytopoiesis and platelet production[J]. Blood, 1991, 77(4): 735-40.
[14] Erslev A J. Megakaryocytic and erythrocytic cell lines share a common precursor cell[J]. Exp Hematol, 1993, 21(10): 1316-1320.
[15] Saxonhouse MA, Rimsza LM, Christensen RD, et al. Effects of anoxia on megakaryocyte progenitors derived from cord blood CD34pos cells[J]. Eur J Haematol, 2003, 71(5): 359-365.
[16] Qi JQ, You T, Pan TT, et al. Erratum to: downregulation of hypoxia-inducible factor-1α contributes to impaired megakaryopoiesis in immune thrombocytopenia[J]. Thromb Haemost, 2017, 117(10): e1-e2.
[17] Lebedeva EV, Yushkov BG, Chereshnev VA. Megakaryocytopoiesis under hypoxic conditions[J]. Bull Exp Biol Med, 2003, 136(6): 554-556.
[18] Nissim Ben Efraim AH, Eliashar R, Levi-Schaffer F. Hypoxia modulates human eosinophil function[J]. Clin Mol Allergy, 2010, 8: 10.
-




 下载:
下载: