Clinicopathological characteristics of histological transformation of non-gastric extranodal mucosa-associated lymphoid tissue lymphoma
-
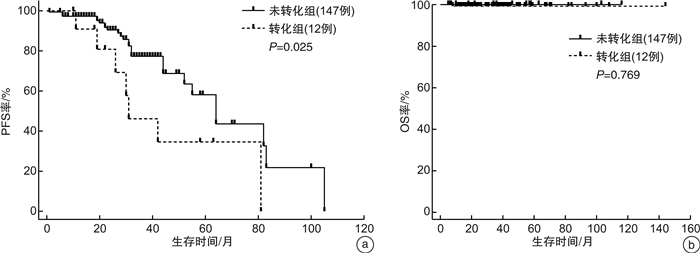
摘要: 目的 探讨非胃型结外黏膜相关淋巴组织淋巴瘤(extranodal mucosa-associated lymphoid tissue,MALT)发生组织学转化(histological transformation,HT)的临床病理特征、影响因素及预后。方法 收集2009年8月—2022年6月诊治的159例非胃型MALT淋巴瘤患者,回顾性分析患者的临床及实验室检查资料,分析其HT的发生率、影响因素及预后。结果 所有患者中位随访时间为24(1~105)个月,34例出现疾病进展或复发,其中1例在确诊8个月后因疾病进展死亡。患者的中位无进展生存期(PFS)及中位总生存期(OS)均未达到,5年OS率为99.4%。患者发生HT的中位年龄为58(23~70)岁,总发生率为7.5%(12/159),5年累积发生率为5.0%。从确诊MALT淋巴瘤到发生HT的中位时间为35(20~81)个月。发生HT的病理类型均为弥漫大B细胞淋巴瘤,其中83.3%(10/12)为非生发中心亚型。临床分期Ⅲ~Ⅳ期(χ2=5.086,P=0.024)、伴有大包块(χ2=7.345,P=0.007)、血清白蛋白 < 35 g/L(χ2=6.470,P=0.011)、血清可溶性白细胞介素2受体(sIL-2R)> 710 U/mL(χ2=6.392,P=0.011)和病理组织Ki67>10%(χ2=13.496,P<0.001)是预测HT高风险的指标。Cox多因素分析结果显示,Ki67>10%是HT的独立影响因素(HR=10.461,95%CI 1.283~85.273,P=0.028)。HT组患者的PFS中位时间31个月,较未HT组PFS中位时间(64个月)明显缩短(P < 0.05)。经过含利妥昔单抗、来那度胺等新药以及CAR-T等新方法的治疗,HT组患者的OS与未HT组比较差异无统计学意义(P>0.05)。结论 非胃型MALT淋巴瘤患者以惰性病程为主,总体预后良好。血清sIL-2R是值得关注的指标,有可能作为HT的生物标志物之一。发生HT后患者PFS明显缩短,及时给予新药和新方法治疗,可能使患者获得长期生存。
-
关键词:
- 结外黏膜相关淋巴组织淋巴瘤 /
- 非胃型 /
- 组织学转化
Abstract: Objective To investigate the clinicopathological features, influencing factors and prognosis of histological transformation(HT) in non-gastric extranodal mucosa-associated lymphoid tissue(MALT) lymphoma.Methods A total of 159 cases of non-gastric MALT lymphoma diagnosed and treated from August 2009 to June 2022 were collected. The clinical data and laboratory examination data of the patients were retrospectively analyzed to analyze the incidence, influencing factors and prognosis of HT.Results The median follow-up time of all patients was 24(1-105) months, and 34 patients developed disease progression or recurrence. Among them, 1 patient died 8 months after diagnosis due to disease progression. The median PFS and median OS were not achieved, and the 5-year OS rate was 99.4%. The median age of HT was 58(23-70) years, the overall incidence was 7.5%(12/159), and the cumulative 5-year incidence was 5.0%. The median time from diagnosis of MALT lymphoma to HT was 35(20-81) months. All the pathological types of HT were DLBCL, and 83.3%(10/12) were non-GCB subtype. Clinical stageⅢ-Ⅳ(χ2=5.086, P=0.024), accompanied by a large mass(χ2=7.345, P=0.007), serum albumin < 35 g/L(χ2=6.470, P=0.011), serum soluble interleukin-2 receptor(sIL-2R) >710 U/mL (χ2=6.392, P=0.011) and pathological tissue Ki67>10%(χ2=13.496, P < 0.001) were high risk predictors of HT. COX multivariate analysis showed that Ki67>10% was an independent influence factor for HT(HR=10.461, 95%CI 1.283-85.273, P=0.028). The median time of PFS in the HT group was 31 months, which was significantly shorter than that in the non-HT group(median time 64 months)(P < 0.05). After treatment with new drugs such as rituximab or lenalidomide and new methods such as CAR-T, there was no statistical difference in OS between HT group and non-HT group(P>0.05).Conclusion Non-gastric MALT lymphoma was characterized by indolent course and good prognosis. Serum sIL-2R was a noteworthy indicator, and may be one of the biomarkers of HT. The PFS of patients with HT was significantly shortened. Timely treatment with new drugs and new methods may help patients to achieve long-term survival. -

-
表 1 初发非胃型MALT淋巴瘤患者及HT患者的临床特征
例(%) 临床特征 全部患者 未转化组 转化组 P 总数 159(100.0) 147(92.5) 12(7.5) 男 62(39.0) 56(38.1) 6(50.0) 0.540 年龄>60岁 62(39.0) 58(39.5) 4(33.3) 0.767 有B症状 18(11.3) 17(11.6) 1(5.6) 1.000 临床分期Ⅲ~Ⅳ期 56(35.2) 48(32.7) 8(66.7) 0.026 MALT-IPI ≥2分 25(15.7) 22(15.0) 3(25.0) 0.404 骨髓受累 46(28.9) 45(30.6) 1(8.3) 0.182 结外器官受累≥2个 56(35.2) 53(36.1) 3(25.0) 0.542 伴大包块 45(28.3) 38(25.9) 7(58.3) 0.040 血红蛋白 < 120 g/L 50(31.4) 47(32.0) 3(25.0) 0.754 血小板 < 100×109/L 16(10.1) 15(10.2) 1(8.3) 1.000 血清LDH>192 U/L 44(27.7) 40(27.2) 4(33.3) 0.739 血清β2-MG>2 366 ng/mL 62(39.0) 56(38.1) 6(50.0) 0.540 血清ALB < 35 g/L 33(20.8) 27(18.4) 6(50.0) 0.019 血清sIL-2R>710 U/mL 73(45.9) 63(42.9) 10(83.3) 0.013 病理组织Ki67>10% 62(39.0) 51(34.7) 11(91.7) <0.001 表 2 初发非胃MALT淋巴瘤患者PFS的影响因素分析
临床相关因素 5年PFS率/% PFS单因素 PFS多因素 χ2 P HR(95%CI) P 是否转化 5.040 0.025 1.509(0.532~4.281) 0.439 否 58.2 是 34.6 一线治疗后疗效 8.102 0.004 2.742(1.127~6.671) 0.026 CR 68.1 未CR 32.1 血清ALB/(g/L) 6.518 0.011 1.360(0.545~3.396) 0.509 ≥35 65.0 < 35 29.1 血清sIL-2R/(U/mL) 3.956 0.047 0.968(0.402~2.334) 0.968 ≤710 71.6 >710 64.4 Ki67增殖指数/% 5.958 0.015 1.617(0.628~4.162) 0.319 ≤10 73.3 >10 39.2 表 3 非胃型MALT淋巴瘤患者HT后的临床特征
编号 性别 年龄/岁 分期 是否伴B症状 IPI评分/分 受累部位 病理类型 转化时的免疫表型 病理组织Ki67/% 转化时的FISH NGS HT后治疗方案 一线方案后疗效 1 女 23 4 否 3 肺、宫颈、胃 non-GCB MUM-1(+),CD10(-),Bcl-6(-),Bcl-2(约20%+),CD30(约30%+),EBER(-) 70 阴性 / R-CEOP;R-ICE SD 2 女 37 4 否 1 扁桃体、鼻咽部 non-GCB MUM-1(大部分+),CD10(-),Bcl-6(-),Bcl-2(部分+),C-MYC(约20%+),EBER(+) 65 IGH分离探针阳性 阴性 未治疗 SD 3 女 38 1 否 1 鼻咽部 non-GCB MUM-1+(70%+),CD10(-),BCL-2(60%),C-MYC(50%+) 60 / / R-CHOP;R-ICE后R维持 CR 4 男 44 1 否 0 腘窝皮肤、腹股沟淋巴结 non-GCB CD10(-),Bcl-2(少量+),EBER(-) 5 IgH分离探针阳性 B2M、PIM1、ID3、ITPKB、CCND3、TP53 放疗 PR 5 男 54 2 否 1 肾脏、腹膜后淋巴结 non-GCB CD10(-),BCL-6(-),Bcl-2(+),BCL-2(+),EBER(-) 50 阴性 TET2、ARID1A、BCLAF1 R-CHOP CR 6 男 57 4 否 2 眼眶、前纵隔、胸椎 non-GCB MUM-1(小部分+),CD10(-),Bcl-6(-),Bcl-2(100%+),c-myc(30%+),EBER(-) 70 阴性 / 大剂量MTX+ICE;放疗;伊布替尼桥接后CAR-T PR 7 男 59 4 是 3 涎腺、腹膜后淋巴结 GCB MUM-1(+),CD10(+),Bcl-6(约60%+),Bcl-2(< 10%+),C-myc(约40%+) 60 BCL6分离探针阳性 / R2-CHOP PR 8 女 60 4 否 2 皮肤、扁桃体、纵隔及腹股沟淋巴结 non-GCB MUM-1(+),CD10(-),Bcl-6(5%+),BCL-2(约100%+),c-myc(约5%+),EBER(-) 35 阴性 CREBBP、ID3、ARID1A、TET2、PRDM1、CARD11、MPEG、CHD2 R2-ICE;CAR-T PD 9 男 62 4 否 3 鼻咽部、多发淋巴结 non-GCB MUM-1(少量+),CD10(-),Bcl-6(-),Bcl-2(-),C-MYC(约2%+),EBER(-) 40 阴性 TET2、TNFAIP3、ITPKB、CIITA R-CHOP CR 10 女 64 3 否 2 眼眶、多发淋巴结 non-GCB Bcl-2(+),Bcl-6(-),CD10(-) 40 阴性 阴性 R-CHOP CR 11 女 67 4 是 3 肝脏、腹膜后淋巴结 non-GCB MUM-1(小部分+),CD10(-),Bcl-6(-),BCL-2(+),C-myc(约8%+),EBER(-) 30 / 阴性 BR CR 12 男 70 4 否 4 颌下腺、脾脏、多发淋巴结 / / / / / R-CHOP CR 表 4 初发非胃MALT淋巴瘤患者HT的影响因素分析
临床相关因素 单因素 多因素 χ2 P HR(95%CI) P 临床分期 5.086 0.024 2.321(0.575~9.378) 0.237 Ⅰ~Ⅱ Ⅲ~Ⅳ 是否伴大包块 7.345 0.007 2.216(0.596~8.239) 0.235 否 是 血清白蛋白/(g/L) 6.470 0.011 2.073(0.642~6.693) 0.223 ≥35 < 35 血清sIL-2R/(U/mL) 6.392 0.011 2.239(0.444~11.291) 0.329 ≤710 >710 Ki67增殖指数/% 13.496 < 0.001 10.461(1.283~85.273) 0.028 ≤10 >10 -
[1] Sriskandarajah P, Dearden CE. Epidemiology and environmental aspects of marginal zone lymphomas[J]. Best Pract Res Clin Haematol, 2017, 30(1-2): 84-91. doi: 10.1016/j.beha.2016.07.002
[2] Nakamura S, Ponzoni M. Marginal zone B-cell lymphoma: lessons from Western and Eastern diagnostic approaches[J]. Pathology, 2020, 52(1): 15-29. doi: 10.3969/j.issn.1006-7299.2020.01.004
[3] Khalil MO, Morton LM, Devesa SS, et al. Incidence of marginal zone lymphoma in the United States, 2001-2009 with a focus on primary anatomic site[J]. Br J Haematol, 2014, 165(1): 67-77. doi: 10.1111/bjh.12730
[4] Hu QL, Zhang YZ, Zhang XY, et al. Gastric mucosa-associated lymphoid tissue lymphoma and Helicobacter pylori infection: a review of current diagnosis and management[J]. Biomark Res, 2016, 4: 15-23. doi: 10.1186/s40364-016-0068-1
[5] Zucca E, Arcaini L, Buske C, et al. Marginal zone lymphomas: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up[J]. Ann Oncol, 2020, 31(1): 17-29. doi: 10.1016/j.annonc.2019.10.010
[6] Chen PJ, El Hussein S, Xing FY, et al. Chronic lymphocytic leukemia progression diagnosis with intrinsic cellular patterns via unsupervised clustering[J]. Cancers(Basel), 2022, 14(10): 2398-2413.
[7] 阿孜古丽·麦合麦提, 陈菲菲, 任雨虹, 等. 288例滤泡性淋巴瘤患者临床特点及预后分析[J]. 临床血液学杂志, 2022, 35(1): 21-28. doi: 10.13201/j.issn.1004-2806.2022.01.005 https://lcxy.whuhzzs.com/article/doi/10.13201/j.issn.1004-2806.2022.01.005
[8] Durot E, Kanagaratnam L, Zanwar S, et al. A prognostic index predicting survival in transformed Waldenström macroglobulinemia[J]. Haematologica, 2021, 106(11): 2940-2946.
[9] Younes A, Hilden P, Coiffier B, et al. International Working Group consensus response evaluation criteria in lymphoma(RECIL 2017)[J]. Ann Oncol, 2017, 28(7): 1436-1447. doi: 10.1093/annonc/mdx097
[10] Zanwar S, Abeykoon JP, Durot E, et al. Impact of MYD88L265Pmutation status on histological transformation of Waldenström Macroglobulinemia[J]. Am J Hematol, 2020, 95(3): 274-281. doi: 10.1002/ajh.25697
[11] Conconi A, Franceschetti S, Aprile von Hohenstaufen K, et al. Histologic transformation in marginal zone lymphomas 2020[J]. Ann Oncol, 2015, 26(11): 2329-2335. doi: 10.1093/annonc/mdv368
[12] 陈青, 杨含金, 滕晓东. 黏膜相关淋巴组织结外边缘区淋巴瘤171例临床病理与预后分析[J]. 临床与实验病理学杂志, 2020, 36(11): 1301-1306. https://www.cnki.com.cn/Article/CJFDTOTAL-LSBL202011010.htm
[13] Bastidas-Mora G, Beà S, Navarro A, et al. Clinico-biological features and outcome of patients with splenic marginal zone lymphoma with histological transformation[J]. Br J Haematol, 2022, 196(1): 146-155. doi: 10.1111/bjh.17815
[14] 赵林俊, 刘芳勋, 王小沛, 等. 胃黏膜相关淋巴组织淋巴瘤患者68例临床预后分析[J]. 中华医学杂志, 2020, 100(32): 2511-2517. doi: 10.3760/cma.j.cn112137-20200511-01504
[15] MaeshimaAM, Taniguchi H, Toyoda K, et al. Clinicopathological features of histological transformation from extranodal marginal zone B-cell lymphoma of mucosa-associated lymphoid tissue to diffuse large B-cell lymphoma: an analysis of 467 patients[J]. Br J Haematol, 2016, 174(6): 923-931. doi: 10.1111/bjh.14153
[16] Gorodetskiy VR, Probatova NA, Radenska-Lopovok SG, et al. Clonal relationship of marginal zone lymphoma and diffuse large B-cell lymphoma in Sjogren's syndrome patients: case series study and review of the literature[J]. RheumatolInt, 2020, 40(3): 499-506. doi: 10.1007/s00296-019-04470-x
[17] van den Brand M, van der Velden WJ, Diets IJ, et al. Clinical features of patients with nodal marginal zone lymphoma compared to follicular lymphoma: similar presentation, but differences in prognostic factors and rate of transformation[J]. Leuk Lymphoma, 2016, 57(7): 1649-1656. doi: 10.3109/10428194.2015.1106535
[18] Komiya I, Tomoyose T, Ouchi G, et al. Low level of serum HDL-cholesterol with increased sIL-2R predicts a poor clinical outcome for patients with malignant lymphoma and adult T-cell leukemia-lymphoma[J]. Cytokine, 2018, 105: 57-62. doi: 10.1016/j.cyto.2018.02.005
[19] Zhong HJ, Chen J, Cheng S, et al. Prognostic nomogram incorporating inflammatory cytokines for overall survival in patients with aggressive non-Hodgkin's lymphoma[J]. EBio Med, 2019, 41: 167-174.
[20] Kameda T, Shide K, Tahira Y, et al. Prognosis of indolent adult T-cell leukemia/lymphoma[J]. Viruses, 2022, 14(4): 710-718. doi: 10.3390/v14040710
[21] Yu K, Xie W, Zhao LL, et al. sIL-2R: anovel diagnostic biomarker for primary adrenal lymphoma[J]. J ClinEndocrinolMetab, 2022, 107(6): e2284-e2290.
[22] Oiwa K, Fujita K, Lee S, et al. Prognostic impact of six versus eight cycles of standard regimen in patients with diffuse large B-cell lymphoma: propensity score-matching analysis[J]. ESMO Open, 2021, 6(4): 100210-100217. doi: 10.1016/j.esmoop.2021.100210
[23] Jin J, Ji DM, Xia ZG, et al. Four cycles of R-CHOP followed by two applications of rituximab based on negative interim PET/CT: an analysis of a prospective trial[J]. BMC Cancer, 2022, 22(1): 403. doi: 10.1186/s12885-022-09486-4
[24] 钟启, 刘梦珂, 秦维, 等. 苯达莫司汀联合利妥昔单抗治疗进展期边缘区淋巴瘤的临床因素分析[J]. 临床血液学杂志, 2022, 35(11): 788-794. https://lcxy.whuhzzs.com/article/doi/10.13201/j.issn.1004-2806.2022.11.007
-




 下载:
下载: